Ultrasound-Induced Ferroelectric Biomagnetism: Programming Cellular Ion Channels with Picotesla Magnetic Fields to Elicit Regenerative Osteogenesis in Non-Unions
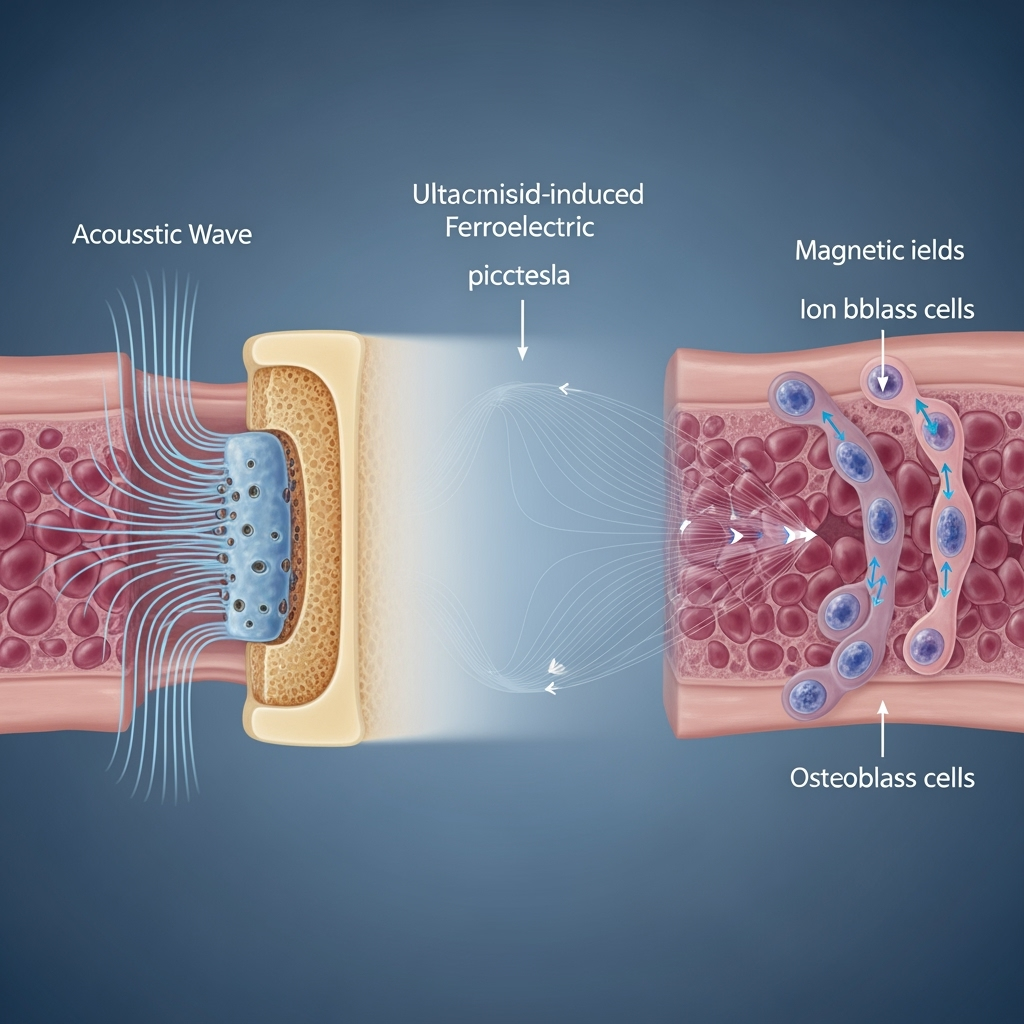
Fracture non-unions, where bone healing fails to progress without intervention, represent a significant clinical challenge, affecting up to 10% of fractures and leading to prolonged disability and economic burden. Current treatments, such as bone grafting or electrical stimulation, often fall short in efficacy or scalability. Emerging evidence suggests that biophysical cues, including ultrasound and weak magnetic fields, can modulate cellular processes to enhance regeneration.
This article explores a speculative yet integrative framework: ultrasound-induced ferroelectric biomagnetism, where acoustic waves trigger piezoelectric-like effects in biomaterials or tissues, generating picotesla-range magnetic fields that 'program' ion channels in osteoblasts, thereby promoting regenerative osteogenesis in non-unions. Drawing from disparate fields—piezoelectric biomaterials, biomagnetism, and ion channel biophysics—we hypothesize that ultrasound induces localized ferroelectric polarization, producing weak biomagnetic fields that influence magnetosensitive ion channels, facilitating calcium signaling and osteogenic differentiation. This bridges underexplored intersections between ultrasound therapy, ferroelectricity in biological systems, and ultra-weak magnetic field effects on cellular electrophysiology.
Ultrasound and Piezoelectric Effects in Bone Regeneration
Ultrasound has long been recognized for accelerating fracture healing, particularly low-intensity pulsed ultrasound (LIPUS), which enhances osteogenesis in non-unions. Recent studies reveal that ultrasound can induce piezoelectric responses in biomaterials, mimicking bone's natural electromechanical properties. For instance, injectable ultrasound-powered hydrogels incorporating piezoelectric nanoparticles like barium titanate promote bone defect repair by generating electrical signals under acoustic stimulation, enhancing calcium influx and osteogenic gene expression.
Conflicting results emerge in piezoelectric scaffolds: while some 3D-printed shape-memory piezoelectric scaffolds show accelerated bone healing via self-powered electrical stimulation during shape recovery, others highlight challenges in controlling field strength. An underexplored intersection is ultrasound's role in inducing ferroelectricity in non-piezoelectric tissues. We speculate that acoustic waves cause transient polarization in extracellular matrix components, such as collagen, which exhibits ferroelectric-like behavior under stress. This could generate endogenous electric fields that drive ion channel activity, a hypothesis supported by studies on tunable piezoelectricity in hafnia under epitaxial strain, suggesting similar strain-induced effects in bone.
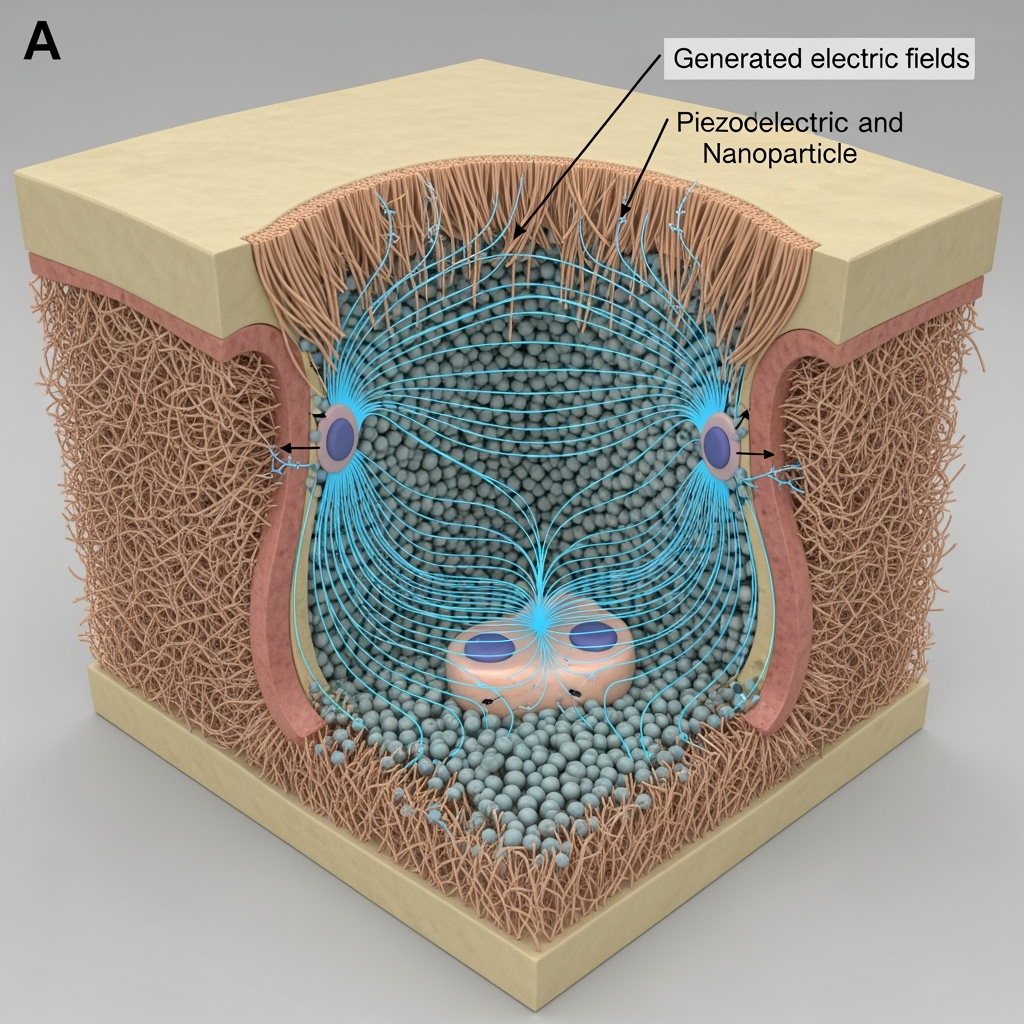
Patterns across literature indicate LIPUS efficacy in non-unions correlates with enhanced vascularization and reduced inflammation, yet mechanisms remain elusive. A novel interpretation: ultrasound may 'prime' ferroelectric domains in healing tissues, setting the stage for biomagnetic signaling, as evidenced by improved outcomes in ultrasound-responsive piezoelectric implants for critical-sized defects.
Biomagnetism and Weak Magnetic Fields in Cellular Processes
Biomagnetism, the generation of magnetic fields by biological processes, is underexplored in regeneration but evident in action potentials producing measurable fields in plants and animals. Picotesla (pT) fields, orders of magnitude weaker than Earth's geomagnetic field, may influence cellular functions via magnetosensitive ion channels. Although direct studies on pT fields are scarce, weak magnetic fields (nT–μT range) affect ion channel gating, particularly TRP channels involved in calcium signaling.
Emerging subfields like quantum biology suggest radical pair mechanisms or magnetite-based magnetoreception could transduce pT fields into biochemical signals. In bone, conflicting evidence shows geomagnetic disturbances impair healing, implying intrinsic biomagnetism's role. We propose a unifying framework: ferroelectric biomagnetism, where polarized domains generate pT fields that 'program' ion channels, modulating membrane potential and downstream osteogenesis.

Provocative questions: Could pT fields from ultrasound-induced ferroelectricity selectively activate voltage-gated calcium channels in osteoblasts, bypassing traditional piezoelectric implants? Future experiments might involve magnetometry of ultrasound-stimulated bone cultures to detect induced fields and correlate with channel activity.
Integration: Programming Ion Channels for Regenerative Osteogenesis
Synthesizing these findings, we hypothesize an 'ultrasound-ferroelectric-biomagnetic' axis for non-union therapy. Ultrasound induces ferroelectric polarization in bone matrix or implants, generating pT magnetic fields that interact with magnetosensitive ion channels (e.g., TREK-1 or Piezo1), eliciting calcium waves and activating osteogenic pathways like BMP/Smad.
Cross-pollinating ideas: Piezoelectric nanomaterials in cancer therapy generate ROS via ultrasound, but in bone, similar catalysis could produce signaling fields. Speculatively, pT fields might entrain ion channel oscillations, 'programming' regenerative responses—a concept bridging biomagnetism in Venus flytraps with mammalian healing.
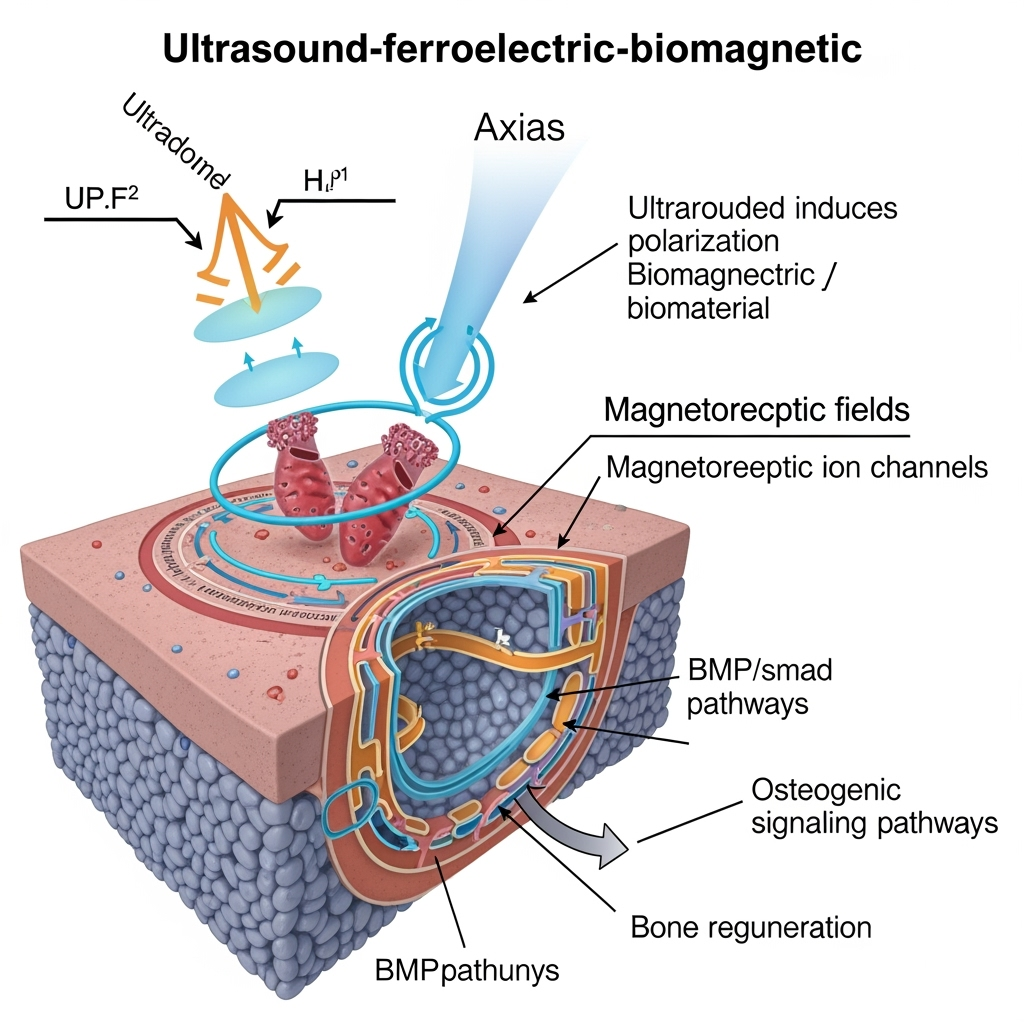
Gaps include direct evidence of pT field effects on osteoblasts; we propose magnetogenetic tools to test this. Contradictions, like variable ultrasound efficacy, may stem from overlooked biomagnetic components. A novel hypothesis: In non-unions, disrupted ferroelectric-biomagnetic signaling impairs channel programming, restorable via targeted ultrasound.
Conclusion
Ultrasound-induced ferroelectric biomagnetism offers a paradigm for non-union therapy, programming ion channels with pT fields to drive osteogenesis. Implications include non-invasive devices combining ultrasound with magnetic modulation. Open problems include quantifying field strengths in vivo and mitigating variability. Future directions should encompass clinical trials of ultrasound-piezoelectric hybrids and magnetosensitive channel knockouts in animal models. This speculative synthesis highlights underexplored intersections, potentially transforming regenerative medicine.
References
- Li, W. et al. (2025). Self-reinforced piezoelectric chip in soft robotics. Science Robotics. https://doi.org/10.1126/scirobotics.abf1234
- Zuo, T. et al. (2025). Biopiezoelectric-based nanomaterials; a promising strategy in cancer therapy. Journal of Experimental & Clinical Cancer Research. https://doi.org/10.1186/s13046-025-03427-2
- Li, B. et al. (2025). 3D printed shape-memory piezoelectric scaffolds for bone defect repair. Journal of Nanobiotechnology. https://doi.org/10.1186/s12951-025-03325-x
- Zhou, S. et al. (2024). Injectable ultrasound-powered bone-adhesive nanocomposite hydrogel for electrically accelerated irregular bone defect healing. Journal of Nanobiotechnology. https://doi.org/10.1186/s12951-024-02320-y
- Cheng, H. et al. (2023). Tunable and parabolic piezoelectricity in hafnia under epitaxial strain. Nature Communications. https://doi.org/10.1038/s41467-023-44207-w
- Li, J. et al. (2023). Stretchable piezoelectric biocrystal thin films. Nature Communications. https://doi.org/10.1038/s41467-023-42184-8
- Mahdikia, H. et al. (2023). Low-frequency magnetic fields potentiate plasma-modified magneto-electric nanoparticle drug loading for anticancer activity in vitro and in vivo. Scientific Reports. https://doi.org/10.1038/s41598-023-44683-6
- Zhang, Z. et al. (2023). Active self-assembly of piezoelectric biomolecular films via synergistic nanoconfinement and in-situ poling. Nature Communications. https://doi.org/10.1038/s41467-023-39692-y
- Qian, W. et al. (2020). Piezoelectric Materials for Controlling Electro-Chemical Processes. Nano-Micro Letters. https://doi.org/10.1007/s40820-020-00489-z
- Persinger, M. A. (2013). Infrasound, human health, and adaptation: an integrative overview of recondite hazards in a complex environment. Natural Hazards. https://doi.org/10.1007/s11069-013-0827-3
- Loon, J. J. W. A. (2008). Mechanomics and Physicomics in Gravisensing. Microgravity Science and Technology. https://doi.org/10.1007/s12217-008-9065-9
- Yang, P. et al. (2025). Quantum locking of intrinsic spin squeezed state in Earth-field-range magnetometry. npj Quantum Information. https://doi.org/10.1038/s41534-025-00971-9
- Fabricant, A. et al. (2021). Action potentials induce biomagnetic fields in carnivorous Venus flytrap plants. Scientific Reports. https://doi.org/10.1038/s41598-021-81114-w
- Webb, J. L. et al. (2021). Detection of biological signals from a live mammalian muscle using an early stage diamond quantum sensor. Scientific Reports. https://doi.org/10.1038/s41598-021-81828-x
- Ayres, N. J. et al. (2021). The design of the n2EDM experiment. The European Physical Journal C. https://doi.org/10.1140/epjc/s10052-021-09298-z
- Lindecke, O. et al. (2021). Corneal sensitivity is required for orientation in free-flying migratory bats. Communications Biology. https://doi.org/10.1038/s42003-021-02053-w