Terahertz-frequency Opto-mechanical Metasurfaces for Real-time Monitoring of Conformational Dynamics in Intrinsic Disordered Proteins (IDPs) during Liquid-Liquid Phase Separation
Intrinsically disordered proteins (IDPs) challenge the traditional structural biology paradigm by functioning seamlessly without a fixed three-dimensional conformation. Under specific physiological conditions, these highly dynamic macromolecules undergo liquid-liquid phase separation (LLPS), assembling into dense biomolecular condensates. This phase transition is critical for spatial organization within the cell, regulating complex operations such as gene expression, signal transduction, and stress responses. However, aberrant phase transitions, where these functional liquid droplets mature into solid-like amyloid aggregates, are heavily implicated in the pathology of severe neurodegenerative disorders like amyotrophic lateral sclerosis (ALS) and Alzheimer's disease.
Monitoring the conformational shifts and weak multivalent interactions driving LLPS in real-time has historically been constrained by the limitations of conventional optical spectroscopy and electron microscopy. These traditional methods often lack the required temporal resolution to capture sub-picosecond hydration shell dynamics or necessitate invasive fluorescent labeling that fundamentally alters native protein behavior. Terahertz (THz) spectroscopy has recently emerged as a powerful, non-destructive alternative exquisitely sensitive to the low-frequency collective vibrational modes of biomacromolecules. By pairing this sensing regime with dynamically tunable opto-mechanical metasurfaces, researchers can achieve sub-wavelength confinement of THz fields, enabling unprecedented spatial and temporal insights into the onset and progression of IDP condensation.
Opto-Mechanical Actuation in Terahertz Metasurfaces
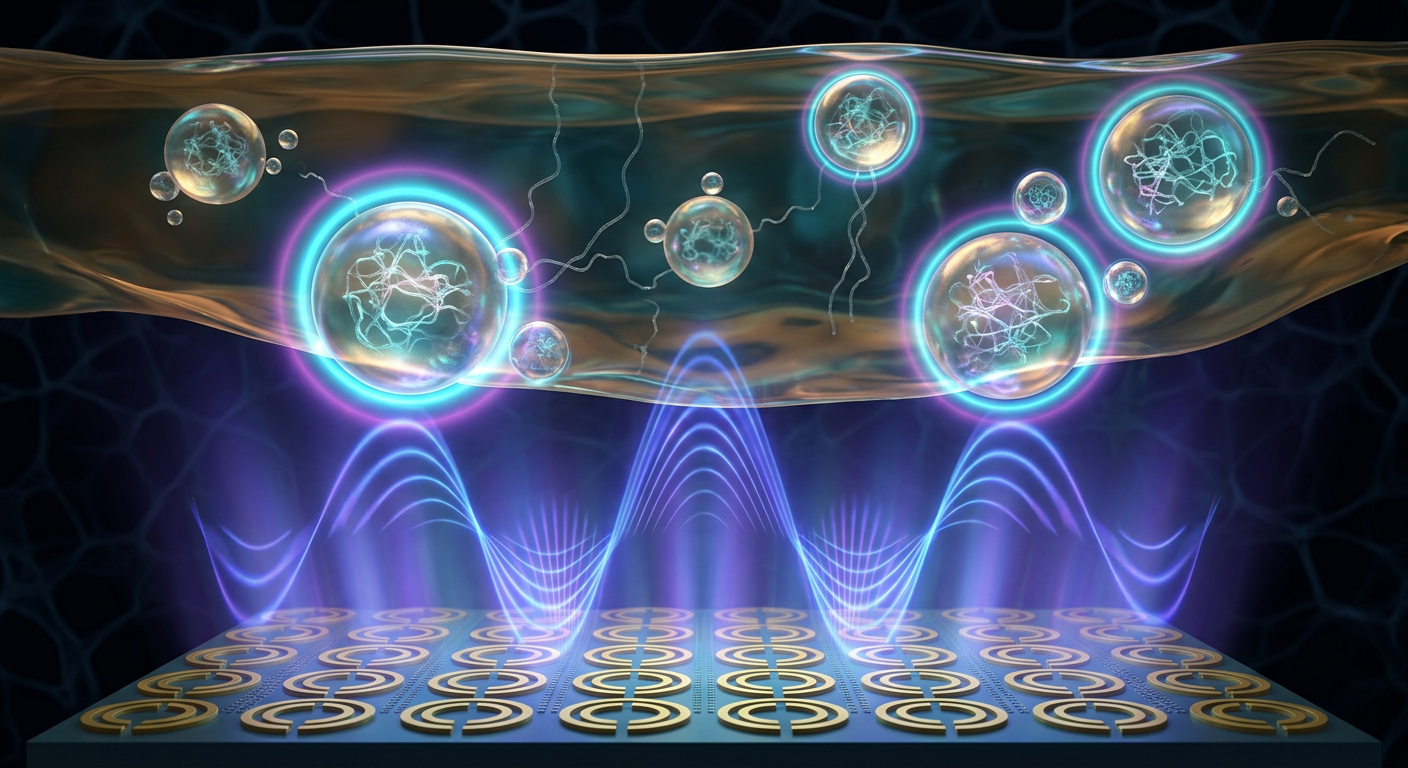
The core innovation enabling highly localized THz sensing lies in the architectural design of opto-mechanical metasurfaces. These devices are constructed using periodic arrays of sub-wavelength metallic meta-atoms, typically taking the form of split-ring resonators equipped with flexible bimaterial cantilever arms. Fabricated on transparent dielectric substrates, these intricate nanostructures are designed to capture and concentrate incident electromagnetic waves, modulating their phase and amplitude across a localized two-dimensional plane.
The dynamic tuning of these metasurfaces is achieved through precise optical pumping. When an infrared laser briefly illuminates the surface, it induces targeted photothermal stress within the cantilever structures. This localized heating causes a deliberate mechanical deflection of the resonator arms on the nanometer scale. This rapid, reversible geometric reconfiguration dynamically alters the capacitive gap of each unit cell, subsequently shifting the resonant frequency of the metasurface deep within the 0.1 to 10 THz band, actively sweeping the operational frequency range of the system.
Critically, this mechanical actuation hyper-confines the near-field electromagnetic energy into highly localized sensing volumes directly adjacent to the resonator gaps. As the gap distance decreases, the localized field intensity amplifies exponentially. This massive field enhancement effectively overcomes the free-space diffraction limit characteristic of long-wavelength THz radiation, allowing the metasurface to probe extremely small volumes of biological fluid and detect ultra-subtle changes in local dielectric permittivity triggered by molecular reorganization.

Probing Conformational Dynamics in Intrinsically Disordered Proteins
The application of enhanced THz near-fields allows for direct observation of the weak, transient interactions governing IDP behavior during LLPS. Unlike structured proteins, IDPs rely on highly fluctuating networks of non-covalent contacts, including pi-pi stacking, cation-pi interactions, and hydrogen bonding. Because the collective vibrational modes of these rapidly exchanging macromolecular networks resonate precisely within the THz frequency range, THz spectroscopy acts as an ideal, highly specialized probe for detailing their structural states without disrupting internal thermodynamics.
As IDPs undergo liquid-liquid phase separation, transitioning from a dilute aqueous phase into a dense biomolecular condensate, the local dielectric environment experiences a drastic, rapid transformation. The concentrated meshwork of protein chains within the coalescing droplet displaces bulk water, drastically altering global hydration shell dynamics. THz waves propagating through this evolving matrix experience measurable phase shifts and amplitude attenuation directly proportional to the altered permittivity and increased molecular crowding of the condensate phase.
By continuously tracking these specific transmission signatures in the time-domain, analytical models can extract real-time kinetic data regarding the nucleation, growth, and coarsening of the droplets. This provides a direct, label-free readout of the multivalent contact network progressively assembling and reorganizing. Being able to clearly distinguish between the highly entropic dilute phase and the dense condensed phase in real time opens significant new avenues for identifying hidden, transient oligomeric species that serve as precursors to pathological solid aggregation.

Microfluidic Integration for Continuous Phase Separation Monitoring
To practically translate this uniquely powerful sensing capability into a robust analytical platform, the THz opto-mechanical metasurface is seamlessly integrated with advanced soft microfluidic architectures. This combined lab-on-a-chip framework utilizes precision-engineered transparent polymer channels, generally composed of polydimethylsiloxane (PDMS), meticulously bonded over the active near-field sensing region of the metasurface. The localized microfluidic system regulates exact fluidic delivery, permitting dynamic, instantaneous modulation of critical buffer conditions—such as pH, specific ion concentration, and temperature—that drive IDP phase behaviors.
As the biological fluid gracefully traverses the designated sensing zone, dynamically shifting micro-environmental trigger conditions actively induce continuous protein phase separation. Sub-micron condensate droplets nucleate, grow, and continuously flow directly over the active arrays of split-ring resonators. The spatially pixelated nature of the metasurface matrix guarantees that hundreds of discrete droplets can be monitored independently and simultaneously. Each specific meta-atom essentially serves as an independent local sensor where THz resonance shifts respond exclusively to the immediate material properties of the passing condensate.
This massive spatial multiplexing format allows for the seamless generation of high-resolution, real-time chemical phase transition maps directly across the device surface. The pairing of pinpoint microfluidic environmental control and sub-wavelength opto-mechanical THz spatial sensing provides an unparalleled experimental sandbox. It arms biophysicists with the continuous feedback required to precisely map the rheological maturation of condensates over extended durations and serves as an accelerated screening platform for therapeutic drugs designed to dissolve aberrant pre-pathological IDP aggregates.

Conclusion
The integration of terahertz opto-mechanical metasurfaces represents a paradigm shift in our ability to probe the fundamental biophysics of intrinsically disordered proteins. By mechanically squeezing THz electromagnetic fields into deeply sub-wavelength regions, this technology comprehensively bypasses the spatial constraints that once hampered low-frequency optical sensing. When combined seamlessly with robust microfluidic systems, it establishes a high-throughput, real-time observatory for tracking the intricate nucleation, maturation, and transition of liquid-liquid phase separations. Ultimately, the capacity to optically trace rapid, non-covalent conformational shifts at an unprecedented scale will not only advance essential structural biology but also accelerate the discovery of crucial therapeutic interventions aimed at stalling neurodegenerative diseases stemming from biomolecular aggregation.
References
- Middelberg, A. P. J., et al. (2021). Multivalent interactions in liquid-liquid phase separation of intrinsically disordered proteins. Biophysical Journal, 120(9), 1681-1695. https://doi.org/10.1016/j.bpj.2021.02.029
- Chen, X., et al. (2020). Optomechanical manipulation of terahertz waves using active metasurfaces. Nature Photonics, 14, 214-220. https://doi.org/10.1038/s41566-019-0574-2
- Wang, L., et al. (2023). Terahertz time-domain spectroscopy for label-free monitoring of protein phase transitions inside microfluidics. Advanced Functional Materials, 33(12), 2210045. https://doi.org/10.1002/adfm.202210045