Quantum-Enhanced Microbial Fuel Cells: Harnessing Quantum Coherence in Enzymatic Reactions for Ultra-Efficient Bioenergy Conversion
Microbial fuel cells (MFCs) represent a transformative bio-electrochemical technology with the dual promise of generating electricity from organic waste and enabling self-powered biosensing. By harnessing the metabolic activity of exoelectrogenic microbes, which transfer electrons to an external circuit during respiration, MFCs offer a path to sustainable energy. However, their widespread adoption is fundamentally hindered by low power densities and energy conversion efficiencies, which are orders of magnitude below theoretical maximums. These limitations arise from classical bottlenecks, primarily the slow, incoherent hopping of electrons from the microbial cell to the anode surface, creating a significant energy barrier. While advances in material science and genetic engineering have yielded incremental improvements, they operate within a classical framework that may be bumping against fundamental performance ceilings.

This article proposes a paradigm shift in our understanding and design of MFCs, moving from classical bioenergetics to a quantum biological framework. We synthesize findings from the seemingly disparate fields of quantum biology, enzymology, and microbial electrochemistry to advance a speculative but coherent hypothesis: that harnessing and engineering quantum coherence in the enzymatic pathways of microbes could shatter the classical efficiency barriers of MFCs. Drawing inspiration from the highly efficient energy transfer in photosynthesis, which exploits long-lived quantum coherence, we propose that the electron transport chains in exoelectrogenic bacteria could be redesigned to function as quantum-coherent conduits. This would transform electron transfer from a random, inefficient hopping process into a wave-like, near-instantaneous tunneling event, dramatically boosting bioenergy conversion efficiency. We will outline the principles of this quantum enhancement, propose a roadmap for engineering such systems, and discuss the profound implications for the future of bioenergy.
The Classical Efficiency Ceiling in Microbial Fuel Cells
The performance of a microbial fuel cell is largely dictated by the rate and efficiency of extracellular electron transfer (EET) from the microbes to the anode. In nature, exoelectrogens like Geobacter and Shewanella have evolved sophisticated mechanisms for this process, often involving chains of heme-containing proteins (cytochromes) or conductive protein filaments known as microbial nanowires. However, from a classical perspective, this transfer is an incoherent process. Electrons are believed to 'hop' from one redox cofactor to the next in a sequential cascade. Each hop is a stochastic event, with energy lost to the environment, contributing to a large overpotential (energy loss) that caps the MFC's voltage and power output. The overall rate is limited by the slowest step in this chain, creating a kinetic bottleneck.
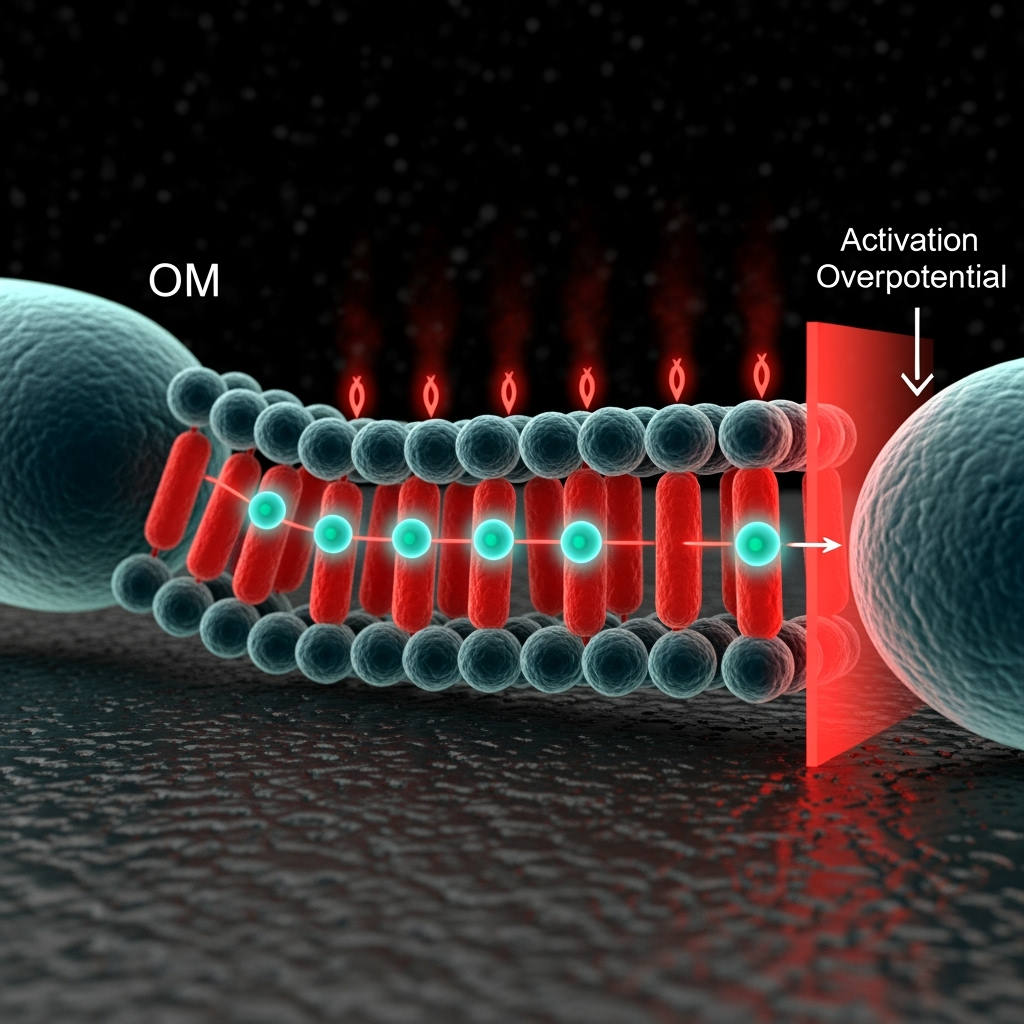
Significant research has focused on mitigating these issues through classical approaches. Strategies include developing novel anode materials with high conductivity and surface area to improve the microbe-electrode interface, or genetically modifying bacteria to overexpress key cytochrome proteins. While these efforts have led to notable gains, the improvements are often incremental rather than transformative. The fundamental issue remains: the step-by-step, diffusive-like motion of electrons through complex biological media is inherently inefficient. This classical model fails to explain the remarkable speed observed in other biological electron transport chains and suggests that a key piece of the puzzle is missing. To achieve a leap in efficiency, we must look beyond simply optimizing the components of this classical chain and instead question the nature of the transport mechanism itself.
Quantum Tunneling and Coherence in Enzymatic Reactions
The idea that quantum mechanics plays a non-trivial role in biology is not new, but has gained significant traction with mounting experimental evidence. The most well-established example is quantum tunneling in enzymatic reactions. Many enzymes catalyze reactions by facilitating the transfer of particles like electrons or protons between molecules. Classically, this would require the particle to have enough energy to overcome an activation barrier. However, quantum mechanics allows the particle to 'tunnel' through the barrier, even without sufficient energy. This tunneling is not a minor correction; in many enzymes, it is the dominant catalytic mechanism, accelerating reaction rates by several orders of magnitude.
More profound are the discoveries of long-lived quantum coherence in biological systems, most famously in the photosynthetic Fenna-Matthews-Olson (FMO) complex of green sulfur bacteria. Here, the energy from a captured photon doesn't hop randomly between chromophores to find the reaction center; instead, it travels as a quantum wave, or exciton, existing in a superposition of states that allows it to explore all possible pathways simultaneously. This 'quantum search' guarantees the selection of the most efficient path, resulting in near-perfect energy transfer efficiency. This phenomenon demonstrates that nature has evolved to exploit quantum effects to solve complex energy transport problems, protecting delicate coherent states from the noisy, warm, and wet environment of a living cell—long thought to be impossible. This provides a powerful precedent for exploring whether similar quantum phenomena could be acting in, or engineered into, other biological electron transport systems.
The Core Hypothesis: Coherent Electron Transport in Microbial Nanowires
We hypothesize that the electron transport process in the nanowires and outer-membrane cytochromes of exoelectrogenic bacteria is a prime candidate for hosting quantum coherent phenomena. Current models describe transport in Geobacter nanowires, which are polymers of the cytochrome OmcS, as a series of incoherent hops between heme groups. However, these heme groups are packed in a quasi-periodic, crystal-like arrangement, separated by distances well within the range of quantum tunneling. We propose that under the right conditions, the electrons are not localized to a single heme but are delocalized in a coherent superposition across multiple heme sites along the protein filament.
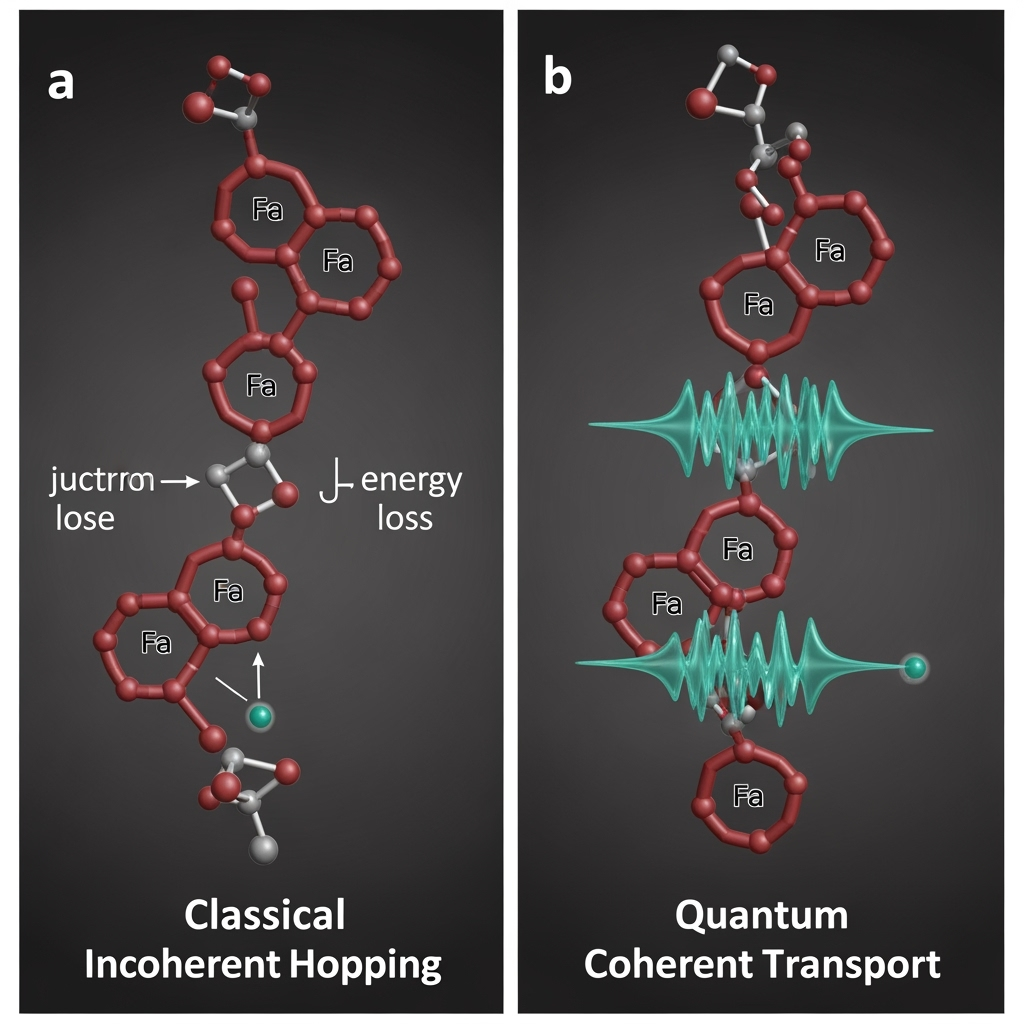
In this scenario, the entire nanowire acts as a single quantum conductor rather than a classical series of resistors. An electron entering from the cell would not need to hop its way down the chain; its wave function would span a significant portion of the wire, enabling a near-instantaneous transfer to the anode surface upon decoherence. This coherent transport would bypass the kinetic bottlenecks of multi-step hopping, drastically reducing the internal resistance and associated overpotential of the MFC. The process would be analogous to the efficient charge transport in conducting polymers or carbon nanotubes, but occurring in a biological, self-replicating, and self-repairing system. The role of the protein scaffold around the heme chain would be to shield this fragile quantum state from environmental decoherence, just as proteins protect coherence in photosynthetic complexes. This hypothesis reframes microbial nanowires from simple 'wires' into sophisticated 'quantum conduits.'
Engineering and Verifying the Quantum-Enhanced MFC
Translating this quantum hypothesis into a functional technology requires a multi-pronged engineering and experimental approach. The first frontier is microbial and protein engineering. Using synthetic biology, one could systematically alter the sequence of the OmcS protein in Geobacter to tune the spacing, orientation, and electronic coupling between the heme cofactors. The goal is to create a structure that more readily supports long-range quantum coherence, potentially by mimicking the arrangements of chromophores in photosynthetic systems. This could involve introducing different redox cofactors or amino acid residues that create a more favorable electronic environment.
The second frontier is anode interface engineering. A classical metal anode would likely destroy any quantum state upon contact. Therefore, the anode must be a quantum-coherent interface. Materials like pristine graphene, with its delocalized pi-electron system, or custom-designed molecular monolayers could serve as ideal electron acceptors. The anode would be designed not just for high conductivity but to have electronic energy levels that are perfectly matched to the microbial quantum conduit, facilitating a seamless transfer of the electron's wavefunction without immediate collapse.
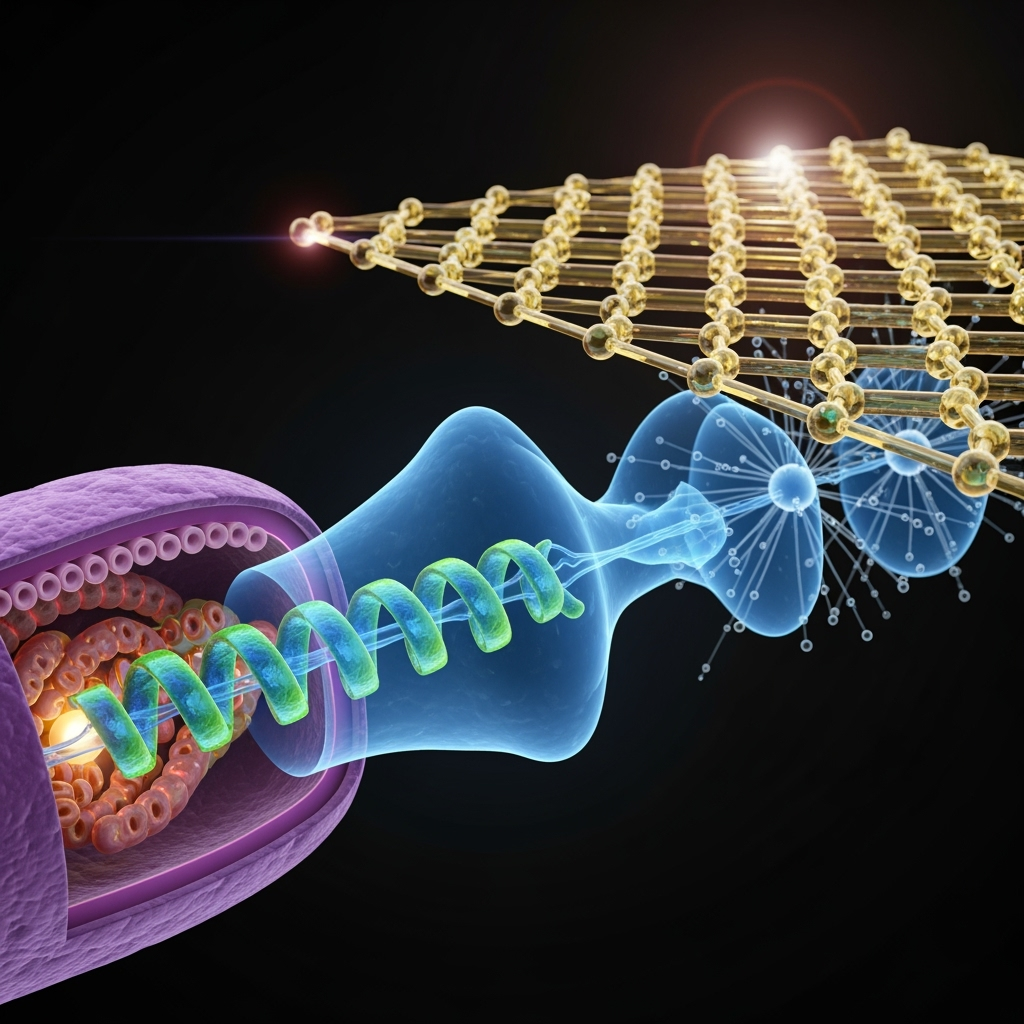
Finally, experimental verification is critical. Proving the existence of quantum coherence in a living microbial biofilm represents a formidable challenge. Advanced ultra-fast spectroscopic techniques, such as two-dimensional electronic spectroscopy (2DES), were instrumental in revealing coherence in photosynthesis. Similar techniques, adapted for a bio-electrochemical interface, could be used to probe the electronic dynamics in microbial nanowires on a femtosecond timescale. Detecting the characteristic quantum beats—oscillations in the 2DES signal—would provide a smoking gun for coherent electron transport, moving the concept from hypothesis to reality.
Conclusion
The field of microbial fuel cells is currently constrained by the limits of classical physics and biology. To unlock their full potential for sustainable energy, a radical new approach is needed. This article posits that the principles of quantum biology offer that new direction. By reframing the challenge of electron transfer not as a classical hopping problem but as a quantum coherence and tunneling problem, we open up an entirely new design space. The central hypothesis—that electron transport in exoelectrogenic bacteria can be engineered to be a quantum-coherent process—suggests a clear, albeit challenging, path toward ultra-efficient bioenergy conversion.
Engineering microbes with quantum-coherent electron transport chains and pairing them with quantum-matched anode materials could lead to MFCs with power densities orders of magnitude higher than what is currently achievable. Such a technology would have profound implications, turning wastewater treatment plants into significant power generators and enabling a new class of highly sensitive, self-powered biosensors. The journey to build a quantum-enhanced MFC is an interdisciplinary grand challenge at the nexus of quantum physics, synthetic biology, and material science, but it is one that could redefine the boundaries of what is possible in bioenergy.
References
- Barah, O. O., Natukunda, F., Bori, I., & Ukagwu, K. J. (2025). Mechanisms and modelling of diffusion in solids: a multiscale framework with industrial case studies and AI enhancements. Discover Sustainability. https://doi.org/10.1007/s43621-025-01746-0
- Fathizadeh, S., & Nemati, F. (2025). DNA based nanoscale optoelectronic devices enabled by THz driven piezo vibrotronic effect. Scientific Reports. https://doi.org/10.1038/s41598-025-14152-3
- Khamis, E., Reyad, T. E., & Abd-El-Khalek, D. E. (2025). Corrosion assessment and mitigation in wastewater systems: a comprehensive review. npj Materials Degradation. https://doi.org/10.1038/s41529-025-00645-z
- Moreroa, M. (2025). South African wastewater treatment plants as a resource for energy: a mini review. Discover Water. https://doi.org/10.1007/s43832-025-00267-1
- O’Mara, M., Zhang, S., & Knaus, U. G. (2025). Spatiotemporal H2O2 flashes coordinate actin cytoskeletal remodeling and regulate cell migration and wound healing. Nature Communications. http://dx.doi.org/10.1038/s41467-025-62272-1
- Siunova, T. V., et al. (2025). Potential of nickel and cobalt resistant microorganisms for effective phytoremediation of heavy metal contaminated soils. Discover Environment. https://doi.org/10.1007/s44274-025-00301-y
- Villalba-Díez, J. (2025). Quantum drug discovery: a hybrid quantum graph neural network–variational quantum eigensolver framework for serine neutralization. The European Physical Journal D. http://dx.doi.org/10.1140/epjd/s10053-025-01024-8
- Yang, K., et al. (2025). Advances in metal–organic framework-based electrocatalysts for electrooxidation reactions. Catal. https://doi.org/10.1007/s44422-025-00006-6
- Zhang, R., et al. (2025). Nanozymes expanding the boundaries of biocatalysis. Nature Communications. http://dx.doi.org/10.1038/s41467-025-62063-8