Ferritin Spin Lattices: Engineering Magnetically Coherent Living Tissues as Spin-Wave Logic Substrates for Biohybrid Information Processing
Biological tissues can host dense, ordered protein assemblies, self-heal, and maintain ionic homeostasis—capabilities that rigid inorganic substrates for magnonics lack. Could we engineer living or living-derived materials to become substrates for spin-wave (magnon) logic? This article proposes ferritin-based spin lattices—protein nanocage arrays loaded with magnetic cores—and magnetosome-inspired biomineral architectures as biohybrid magnonic media. We synthesize evidence from ferritin/magnetosome biophysics, protein self-assembly, and modern magnonics to chart a route to magnetically coherent, low-power information processing embedded in soft, biological environments.
A core premise is to trade ultra-low damping of crystalline yttrium iron garnet (YIG) for biotic advantages (growth, repair, adaptability) by exploiting collective dipolar-coupled nanomagnet arrays, hybrid magnon–phonon transduction, and interference-based logic. We outline architectures that are physically plausible, biologically integrable, and experimentally testable within near-term constraints on magnetic losses and field amplitudes in tissue-like media.

Magnetic building blocks in biology: from ferritin to magnetosomes
Ferritin nanocages (24-mer, ~12 nm outer diameter) mineralize iron as ferrihydrite-like cores that are typically superparamagnetic at room temperature. Their cores possess intrinsic enzyme-mimetic reactivity and antioxidant nanozyme activity, with composition-dependent properties across kingdoms (iron/phosphate ratio governs catalytic behavior) and excellent biocompatibility and modifiability for targeting and trafficking in mammalian cells. Heavy-chain ferritin can be repurposed as a drug/ligand carrier and as a genetically addressable scaffold. Implication: native ferritin cores are not ferromagnetic at room temperature, but the protein shell is a robust, engineerable lattice unit for templating magnetic phases.
Magnetoferritin and encapsulin/engineered nanocages can host ferrimagnetic magnetite or cobalt ferrite, enhancing saturation magnetization and enabling blocked single-domain behavior at room temperature. Encapsulin-produced iron oxide nanocomposites can reach high saturation magnetization and even large specific absorption rates under alternating fields, though heating is neither necessary nor desirable for logic. Implication: genetic cages provide a route to uniform nanomagnets with biological control over core phase and size, and to co-localized catalytic or signaling functions.
Magnetotactic bacteria (MTB) biosynthesize magnetite magnetosomes (~30–50 nm) arranged in chains, forming room-temperature single-domain dipoles with coherent magnetic behavior. Recent surveys map MTB diversity and magnetosome gene clusters across phyla, while efforts to port core magnetosome proteins (e.g., MamI/MamL) into mammalian cells show protein interactions and punctate co-localization. Magnetosomes inside human tumor models show oxidation dynamics and crosstalk with ferritin pools. Giant magnetofossils and MTB ecophysiology underscore the robustness of biogenic magnetic order in varied environments. Implication: while full magnetosome organelles in eukaryotes remain challenging, partial genetic programs and hybrid microbial–materials strategies can provide biologically assembled single-domain arrays in soft matrices.
From nanoparticles to spin lattices: protein-directed assembly and coupling
Spin waves in continuous low-damping films (e.g., YIG) propagate over 10–100 μm at GHz, but protein-caged magnetic nanoparticles are discrete. The goal is not to mimic continuous films, but to build magnonic metamaterials: ordered arrays of dipolar-coupled nanomagnets that support collective modes, bandgaps, and interference—i.e., magnonic crystals.
Engineered ferritin and other protein cages can be induced to form 2D/3D crystals via pH-tunable histidine motifs and metal-mediated crosslinks, yielding prescribed lattice constants and symmetries. Electrostatic co-crystallization of biomacromolecules demonstrates room-temperature aqueous assembly of ordered superlattices and modular post-functionalization. Genetic ferritin variants support programmable self-assembly in cells and ex vivo, and can be orthogonally decorated with ECM-binding peptides to anchor arrays in hydrogels. With 6–10 nm cores and ~12 nm center-to-center spacing, dipolar fields in dense ferritin arrays can reach tens of mT locally, sufficient to generate collective precession modes under a modest bias field. While exchange coupling across protein shells is negligible, dipolar-coupled nanodot arrays are a standard route to magnonic functionality (bandgaps, directional couplers, logic) in inorganic systems. Strategies to enhance coherence include loading cores with magnetite or cobalt ferrite to increase μ0Ms; aligning uniaxial anisotropy via bias fields during assembly; patterning lattice anisotropy (antidot-like scaffolds); and introducing chiral asymmetry to emulate Dzyaloshinskii–Moriya-like nonreciprocity via structured heavy-metal overlays in hybrid devices.
Speculative but testable idea: deposit ferritin lattices atop thin YIG waveguides; use near-field coupling to transduce low-loss YIG magnons into and out of discrete ferritin arrays acting as local processors (filters, majority gates), while retaining long-range communication in YIG. This partitions propagation (YIG) from computation (bio-lattice), easing damping constraints in the biotic medium.

Magnonic functionality targets and constraints in soft media
Loss and dispersion: YIG thin films achieve sub-10−4 damping and >100 μm decay lengths in nanoscale waveguides; magnetite/cobalt ferrite nanodots exhibit higher damping, suggesting sub-micron decay for GHz spin waves. Therefore, logic primitives should emphasize near-field interference and localized resonant processing rather than long-distance transport.
Interference and logic: Demonstrated in YIG are single-shot interference and phase-controlled XNOR gates; reconfigurable couplers and inverse-designed magnonic devices; amplification via pulsed spin–orbit torque; high-data-rate transmitters; and magnetic-field sensors using interference. Translating to ferritin lattices implies operating in the magnetostatic regime (hundreds of MHz to few GHz), using bias fields (5–50 mT) to tune mode spectra, and designing unit cells for robust constructive/destructive interference over a few lattice periods.
Reconfigurability: State writing can be achieved electrically in hybrid devices (spin–orbit torques, current-written nanostates) or by localized magnetic bias from flexible, reprogrammable micro-magnets embedded in hydrogels. Protein lattices offer chemical reconfigurability: pH/ion-switchable crosslinks to alter lattice symmetry and bandgaps, or photo-activated metal coordination to rewire networks.
Hybrid magnon–phonon pathways for tissue-scale signaling
Magnetoelastic coupling offers a path to bridge lossy magnons in soft media with low-loss acoustic waves in hydrated tissues. Strong magnon–phonon coupling has been observed in nanomagnets and is a design theme for hybrid crystals.
In biohybrid substrates, local ferritin arrays act as magnonic resonators that convert to phonons via magnetostriction; collagen/gel matrices serve as phonon waveguides over millimeter distances; remote arrays reconvert phonons to magnons. Cavity-magnon-polariton concepts can be adapted to planar microstrip cavities integrated with hydrogel-embedded lattices to coherently route energy; dynamic Hall-like deflection of cavity magnon–polaritons suggests phase-encoded logic at the network level.
Testable hypothesis: In hydrated hydrogels (sound speed ~1.5 km/s), magnetoelastic conversion near 100–500 MHz can deliver phonon-mediated phase-coherent signals over millimeters with <10 dB loss, enabling biologically relevant spatial scales, while the magnetic subsystems remain local and low-power.
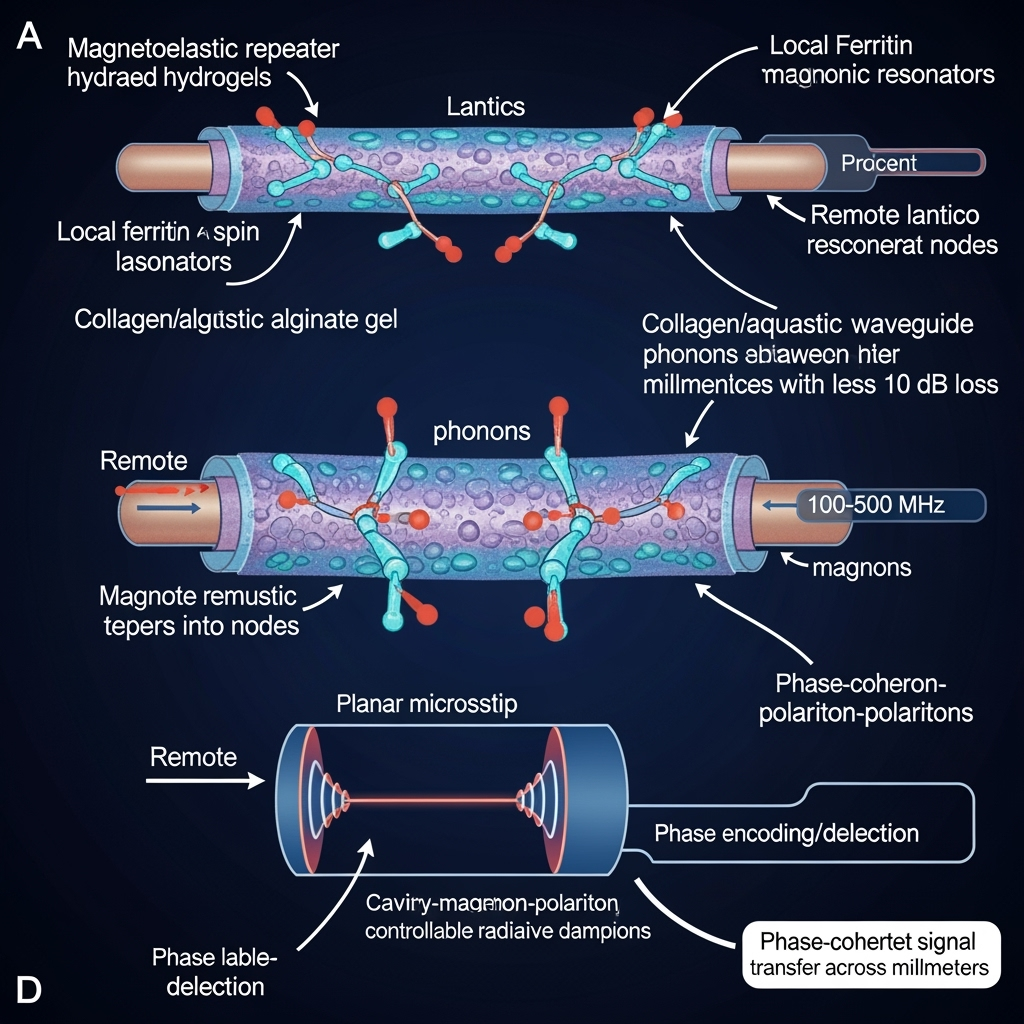
Excitation and readout in and near living tissues
Inductive micro-antennas: Lumped models quantify added inductance from spin-wave excitation; narrow coplanar strips above lattices can excite magnetostatic modes with minimal heating. Antenna scaling and impedance matching at sub-GHz mitigate dielectric loss in tissue.
NV-diamond magnetometry: Single-spin NV sensors image spin waves and spin noise at ~50 nm stand-off, offering non-contact mapping of magnonic fields in vitro; widefield NV layers on diamond chips can interface with thin hydrogel constructs.
Cavity coupling and radiative damping control: Engineering the local density of photon states allows coherent control of magnon radiative damping, enabling tunable coupling between arrays and microwave waveguides.
Electric-field control: Synthetic multiferroic stacks (piezoelectric + magnetostrictive films) modulate spin-wave propagation at low voltages; in soft systems, embedded piezoelectric microbeads or thin PMN-PT chips can modulate local anisotropy for gating.
Biofabrication strategies for magnetically coherent tissues
Genetic nanocages: Express secreted heavy-chain ferritin fused to ECM-binding tags (collagen-binding peptide) to nucleate 2D/3D lattices ex vivo; re-mineralize with magnetite/cobalt ferrite under controlled chemistry. Use histidine motif designs for pH/metal-ion programmable assembly.
Magnetosome-inspired modules: Introduce subsets of magnetosome genes (e.g., MamI/MamL membrane sculptors) to organize intracellular vesicles; co-culture with magnetotactic bacteria or use bacterial cellulose matrices decorated with magnetite via engineered amyloids to form magnetic sheets with precise microstructure.
Hybrid laminates: Stack a YIG nanomembrane (or other low-damping ferrimagnet) beneath a hydrogel with ferritin lattices to implement processor-on-transport separation.
Risk and safety controls
Iron homeostasis: Ferritin nanozymes can scavenge superoxide; surface passivation (dextran/PEG) reduces Fe ion release and ROS; monitor iron-related gene expression and oxidative markers.
Heating and exposure: Operate in low-amplitude rf magnetic fields (μT–mT) and sub-GHz to low-GHz bands to avoid tissue heating; avoid magnetothermal paradigms criticized in magnetogenetics by prioritizing direct magnetic torque and magnetoelastic transduction.
A targeted experimental program
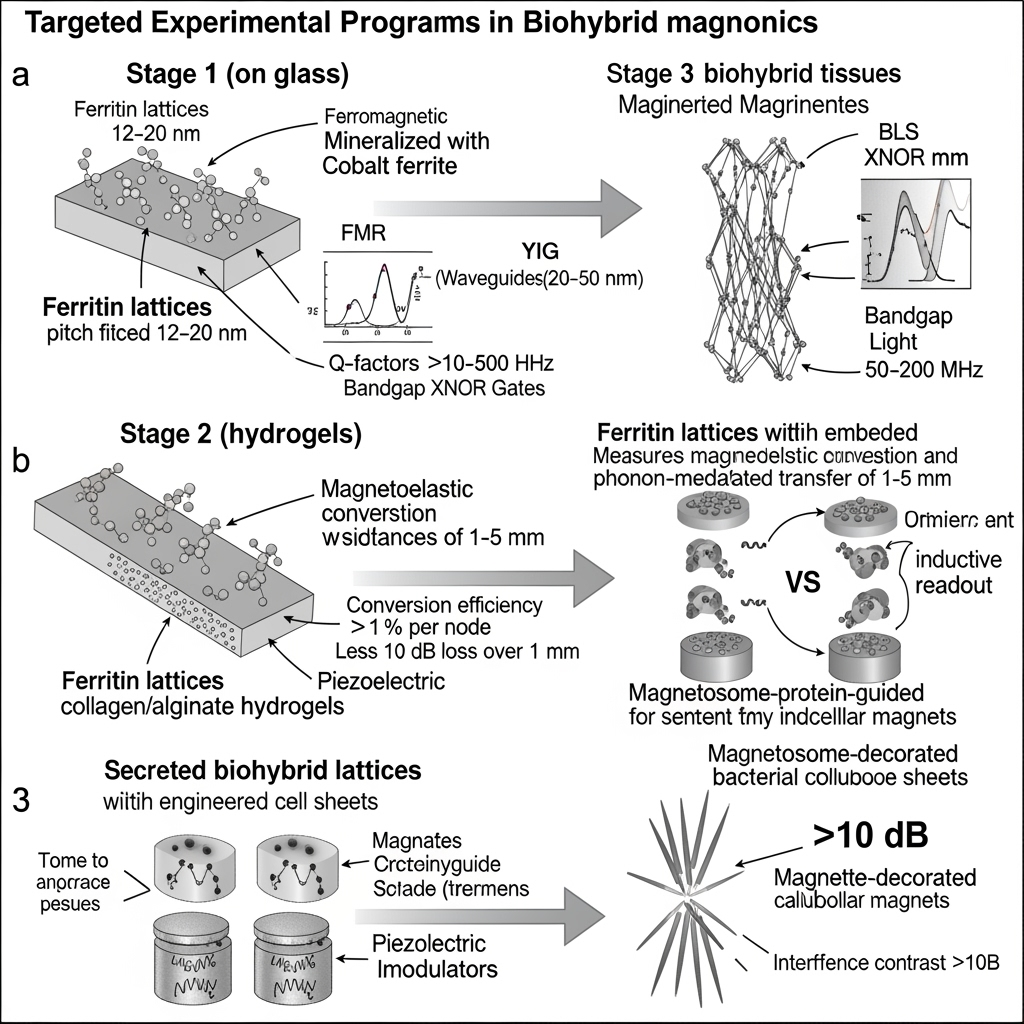
Stage 1: Fabricate ferritin lattices (12–20 nm pitch) on glass; mineralize cores with magnetite and cobalt ferrite; map FMR and BLS spectra; quantify bandgaps and interference at 0.1–3 GHz. Validate dipolar coupling via angle-dependent spectra and micromagnetics; place lattices over 20–50 nm YIG films to demonstrate in–out coupling between YIG waveguides and ferritin metamaterials (filters, XNOR/majority-like functions) at μW–mW rf.
Stage 2: Embed lattices in collagen/alginate hydrogels; measure magnetoelastic conversion efficiency and phonon-mediated transfer between two lattices separated by 1–5 mm; quantify phase fidelity and bit error rates at 10–100 kbps; integrate piezoelectric modulators for electric-field gating of anisotropy.
Stage 3: Express secreted ferritin lattices in engineered cell sheets; assess mechanical and optical integrity; use NV-diamond and inductive probes to map local magnonic fields in vitro; explore magnetosome protein modules (MamI/MamL) to nucleate intracellular magnetic compartments and compare with exogenous magnetite-decorated bacterial cellulose sheets for layered architectures. Key quantitative targets: local resonance Q > 10 at 100–500 MHz in ferritin lattices with magnetite cores; controllable bandgap ~50–200 MHz via lattice reconfiguration; magnetoelastic conversion efficiency > 1% per node; phonon-mediated transfer loss < 10 dB over 1 mm at 200–500 MHz; XNOR-like interference contrast > 10 dB.
Conceptual originality and open questions
Biological coherence without continuous crystals: By embracing discrete dipolar-coupled arrays and magnon–phonon hybrids, biological materials can host wave-based logic without requiring YIG-like perfection. Metabolism as a control channel: Protein-lattice assembly and disassembly (pH, ions, redox) offers reconfigurability unavailable to inorganics.
Open questions: What damping and inhomogeneous broadening arise in ferritin magnetite cores at scale? Can chiral protein lattices emulate DMI-like effects for nonreciprocal spin transport? How does the immune milieu modulate lattice integrity and magnetic noise in vivo? What are the ultimate limits of NV-based readout through hydrated tissues?
Conclusion
Ferritin spin lattices and magnetosome-inspired architectures can act as magnetically coherent processors embedded in soft, living materials if we pivot from long-range, low-loss magnon transport to localized, interference-rich magnonic metamaterials, and bridge nodes with phonons. Protein-directed assembly provides nanoscale order and biological integration; hybrid YIG–bio platforms can deliver high-fidelity transport. A staged program—starting on glass, moving to hydrogels, then to engineered tissues—can test the central hypotheses with established magnonics and biofabrication tools. The prize is a new class of biohybrid substrates where information flows as spin and strain waves, coexisting with growth, healing, and biochemical function.
References
- Ma, L., Zheng, J.-J., Zhou, N., Zhang, R., Fang, L., Yang, Y., Gao, X., Chen, C., Yan, X., & Fan, K. (2023). A natural biogenic nanozyme for scavenging superoxide radicals. Nature Communications. https://doi.org/10.1038/s41467-023-44463-w
- Lee, N. K., Cho, S., & Kim, I.-S. (2022). Ferritin – a multifaceted protein scaffold for biotherapeutics. Experimental & Molecular Medicine. https://doi.org/10.1038/s12276-022-00859-0
- Ravishankar, S., Nedumaran, A. M., Gautam, A., Ng, K. W., Czarny, B., & Lim, S. (2022). Protein nanoparticle cellular fate and responses in murine macrophages. NPG Asia Materials. https://doi.org/10.1038/s41427-022-00453-w
- Volatron, J., Kolosnjaj-Tabi, J., Javed, Y., Vuong, Q. L., Gossuin, Y., Neveu, S., Luciani, N., Hémadi, M., Carn, F., Alloyeau, D., & Gazeau, F. (2017). Physiological Remediation of Cobalt Ferrite Nanoparticles by Ferritin. Scientific Reports. https://doi.org/10.1038/srep40075
- Ducasse, R., Wang, W.-A., Garcia-Jove Navarro, M., Debons, N., Colin, A., Gautier, J., Guigner, J.-M., Guyot, F., & Gueroui, Z. (2017). Programmed Self-Assembly of a Biochemical and Magnetic Scaffold to Trigger and Manipulate Microtubule Structures. Scientific Reports. https://doi.org/10.1038/s41598-017-10297-y
- Maity, B., Abe, S., & Ueno, T. (2017). Observation of gold sub-nanocluster nucleation within a crystalline protein cage. Nature Communications. https://doi.org/10.1038/ncomms14820
- Tan, X., Chen, H., Gu, C., Zang, J., Zhang, T., Wang, H., & Zhao, G. (2020). Converting histidine-induced 3D protein arrays in crystals into their 3D analogues in solution by metal coordination cross-linking. Communications Chemistry. https://doi.org/10.1038/s42004-020-00394-x
- Liljeström, V., Mikkilä, J., & Kostiainen, M. A. (2014). Self-assembly and modular functionalization of three-dimensional crystals from oppositely charged proteins. Nature Communications. https://doi.org/10.1038/ncomms5445
- Zhang, T., & Pan, Y. (2018). Constraining the magnetic properties of ultrafine- and fine-grained biogenic magnetite. Earth, Planets and Space. https://doi.org/10.1186/s40623-018-0978-2
- Carvallo, C., Hickey, S., Faivre, D., & Menguy, N. (2003). Formation of magnetite in Magnetospirillum gryphiswaldense studied with FORC diagrams. Earth, Planets and Space. https://doi.org/10.1186/BF03352894
- Kumar, P., Bulk, M., Webb, A., van der Weerd, L., Oosterkamp, T. H., Huber, M., & Bossoni, L. (2016). A novel approach to quantify different iron forms in ex-vivo human brain tissue. Scientific Reports. https://doi.org/10.1038/srep38916
- Ji, R., Wan, J., He, K., Goswami, P., Shen, J., Pan, Y., & Lin, W. (2025). Magnetosome gene cluster containing bacteria in oxygen-stratified freshwater ecosystems of northern landscapes. Communications Earth & Environment. https://doi.org/10.1038/s43247-025-02495-8
- Pei, Z., Ringe, E., Chang, L., Harrison, R. J., Xue, P., & Williams, W. (2025). Three-dimensional structure, crystallography, and magnetism of giant magnetofossils. Communications Earth & Environment. https://doi.org/10.1038/s43247-025-02396-w
- Sun, Q., Yu, L., Donnelly, S. C., Fradin, C., Thompson, R. T., Prato, F. S., & Goldhawk, D. E. (2024). Essential magnetosome proteins MamI and MamL from magnetotactic bacteria interact in mammalian cells. Scientific Reports. https://doi.org/10.1038/s41598-024-77591-4
- Gubieda, A. G., Gandarias, L., Pósfai, M., Pattammattel, A., Fdez-Gubieda, M. L., Abad-Díaz-de-Cerio, A., & García-Prieto, A. (2024). Temporal and spatial resolution of magnetosome degradation at the subcellular level in a 3D lung carcinoma model. Journal of Nanobiotechnology. https://doi.org/10.1186/s12951-024-02788-8
- Shen, J., Paterson, G. A., Wang, Y., Kirschvink, J. L., Pan, Y., & Lin, W. (2023). Renaissance for magnetotactic bacteria in astrobiology. The ISME Journal. https://doi.org/10.1038/s41396-023-01495-w
- Vadanan, S. V., Pasula, R. R., Joshi, N., & Lim, S. (2024). Bioengineering approach for the design of magnetic bacterial cellulose membranes. Communications Materials. https://doi.org/10.1038/s43246-024-00562-9
- Bensmann, J., Schmidt, R., Nikolaev, K. O., Raskhodchikov, D., Choudhary, S., Bhardwaj, R., Taheriniya, S., Varri, A., Niehues, S., El Kadri, A., Kern, J., Pernice, W. H. P., Demokritov, S. O., Demidov, V. E., Michaelis de Vasconcellos, S., & Bratschitsch, R. (2025). Dispersion-tunable low-loss implanted spin-wave waveguides for large magnonic networks. Nature Materials. https://doi.org/10.1038/s41563-025-02282-y
- Wang, Q., Chumak, A. V., & Pirro, P. (2021). Inverse-design magnonic devices. Nature Communications. https://doi.org/10.1038/s41467-021-22897-4
- Goto, T., Yoshimoto, T., Iwamoto, B., Shimada, K., Ross, C. A., Sekiguchi, K., Granovsky, A. B., Nakamura, Y., Uchida, H., & Inoue, M. (2019). Three port logic gate using forward volume spin wave interference in a thin yttrium iron garnet film. Scientific Reports. https://doi.org/10.1038/s41598-019-52889-w
- Song, M., Polakovic, T., Lim, J., Cecil, T. W., Pearson, J., Divan, R., Kwok, W.-K., Welp, U., Hoffmann, A., Kim, K.-J., Novosad, V., & Li, Y. (2025). Single-shot magnon interference in a magnon-superconducting-resonator hybrid circuit. Nature Communications. https://doi.org/10.1038/s41467-025-58482-2
- Merbouche, H., Divinskiy, B., Gouéré, D., Lebrun, R., El Kanj, A., Cros, V., Bortolotti, P., Anane, A., Demokritov, S. O., & Demidov, V. E. (2024). True amplification of spin waves in magnonic nano-waveguides. Nature Communications. https://doi.org/10.1038/s41467-024-45783-1
- Xue, K., & Victora, R. H. (2024). High data rate spin-wave transmitter. Scientific Reports. https://doi.org/10.1038/s41598-024-73957-w
- Nishitani, Y., Kaneko, Y., & Sekiguchi, K. (2025). Magnetic field detection with single mode spin wave interference in asymmetric structure. Scientific Reports. https://doi.org/10.1038/s41598-025-89367-5
- Vanderveken, F., Tyberkevych, V., Talmelli, G., Sorée, B., Ciubotaru, F., & Adelmann, C. (2022). Lumped circuit model for inductive antenna spin-wave transducers. Scientific Reports. https://doi.org/10.1038/s41598-022-07625-2
- Frey, P., Nikitin, A. A., Bozhko, D. A., Bunyaev, S. A., Kakazei, G. N., Ustinov, A. B., Kalinikos, B. A., Ciubotaru, F., Chumak, A. V., Wang, Q., Tiberkevich, V. S., Hillebrands, B., & Serga, A. A. (2020). Reflection-less width-modulated magnonic crystal. Communications Physics. https://doi.org/10.1038/s42005-020-0281-y
- Ren, Z., Liu, S., Jin, L., Wen, T., Liao, Y., Tang, X., Zhang, H., & Zhong, Z. (2019). Reconfigurable nanoscale spin-wave directional coupler using spin-orbit torque. Scientific Reports. https://doi.org/10.1038/s41598-019-43597-6
- Liao, L., Liu, J., Puebla, J., Shao, Q., & Otani, Y. (2024). Hybrid magnon-phonon crystals. npj Spintronics. https://doi.org/10.1038/s44306-024-00052-1
- Berk, C., Jaris, M., Yang, W., Dhuey, S., Cabrini, S., & Schmidt, H. (2019). Strongly coupled magnon–phonon dynamics in a single nanomagnet. Nature Communications. https://doi.org/10.1038/s41467-019-10545-x
- Rao, J. W., Kaur, S., Yao, B. M., Edwards, E. R. J., Zhao, Y. T., Fan, X., Xue, D., Silva, T. J., Gui, Y. S., & Hu, C.-M. (2019). Analogue of dynamic Hall effect in cavity magnon polariton system and coherently controlled logic device. Nature Communications. https://doi.org/10.1038/s41467-019-11021-2
- Yao, B., Yu, T., Gui, Y. S., Rao, J. W., Zhao, Y. T., Lu, W., & Hu, C.-M. (2019). Coherent control of magnon radiative damping with local photon states. Communications Physics. https://doi.org/10.1038/s42005-019-0264-z
- van der Sar, T., Casola, F., Walsworth, R., & Yacoby, A. (2015). Nanometre-scale probing of spin waves using single electron spins. Nature Communications. https://doi.org/10.1038/ncomms8886
- Wang, X., Xiao, Y., Liu, C., Lee-Wong, E., McLaughlin, N. J., Wang, H., Wu, M., Wang, H., Fullerton, E. E., & Du, C. R. (2020). Electrical control of coherent spin rotation of a single-spin qubit. npj Quantum Information. https://doi.org/10.1038/s41534-020-00308-8
- Alizadeh, N., & Salimi, A. (2021). Multienzymes activity of metals and metal oxide nanomaterials: applications from biotechnology to medicine and environmental engineering. Journal of Nanobiotechnology. https://doi.org/10.1186/s12951-021-00771-1
- Zhang, R., Yan, X., Gao, L., & Fan, K. (2025). Nanozymes expanding the boundaries of biocatalysis. Nature Communications. https://doi.org/10.1038/s41467-025-62063-8
- Mohammadi, M. R., Malkovskiy, A. V., Jothimuthu, P., Kim, K.-M., Parekh, M., Inayathullah, M., Zhuge, Y., & Rajadas, J. (2018). PEG/Dextran Double Layer Influences Fe Ion Release and Colloidal Stability of Iron Oxide Nanoparticles. Scientific Reports. https://doi.org/10.1038/s41598-018-22644-8
- Joudeh, N., & Linke, D. (2022). Nanoparticle classification, physicochemical properties, characterization, and applications: a comprehensive review for biologists. Journal of Nanobiotechnology. https://doi.org/10.1186/s12951-022-01477-8
- Ahmed, M. H., Jeske, J., & Greentree, A. D. (2017). Guided magnonic Michelson interferometer. Scientific Reports. https://doi.org/10.1038/srep41472
- Flores-Farías, J., Gallardo, R. A., Brevis, F., Roldán-Molina, A., Cortés-Ortuño, D., & Landeros, P. (2022). Omnidirectional flat bands in chiral magnonic crystals. Scientific Reports. https://doi.org/10.1038/s41598-022-20539-3
- Balinskiy, M., Chavez, A. C., Barra, A., Chiang, H., Carman, G. P., & Khitun, A. (2018). Magnetoelectric Spin Wave Modulator Based On Synthetic Multiferroic Structure. Scientific Reports. https://doi.org/10.1038/s41598-018-28878-w
- Divinskiy, B., Merbouche, H., Demidov, V. E., Nikolaev, K. O., Soumah, L., Gouéré, D., Lebrun, R., Cros, V., Ben Youssef, J., Bortolotti, P., Anane, A., & Demokritov, S. O. (2021). Evidence for spin current driven Bose-Einstein condensation of magnons. Nature Communications. https://doi.org/10.1038/s41467-021-26790-y
- Ren, H., Zheng, X. Y., Channa, S., Wu, G., O’Mahoney, D. A., Suzuki, Y., & Kent, A. D. (2023). Hybrid spin Hall nano-oscillators based on ferromagnetic metal/ferrimagnetic insulator heterostructures. Nature Communications. https://doi.org/10.1038/s41467-023-37028-4
- Furukawa, R., Nezu, S., Eguchi, T., & Sekiguchi, K. (2023). Mode-dependent magnonic noise. NPG Asia Materials. https://doi.org/10.1038/s41427-023-00522-8
- Wang, Q., Verba, R., Davídková, K., Heinz, B., Tian, S., Rao, Y., Guo, M., Guo, X., Dubs, C., Pirro, P., & Chumak, A. V. (2024). All-magnonic repeater based on bistability. Nature Communications. https://doi.org/10.1038/s41467-024-52084-0