Biogenic Cold-Weld Carbon Fixation: Lithifying Atmospheric CO₂ into Maraging-Steel-Strength Biocement via Enzymatic Metal Coordination Networks in Alkaliphilic Microbes
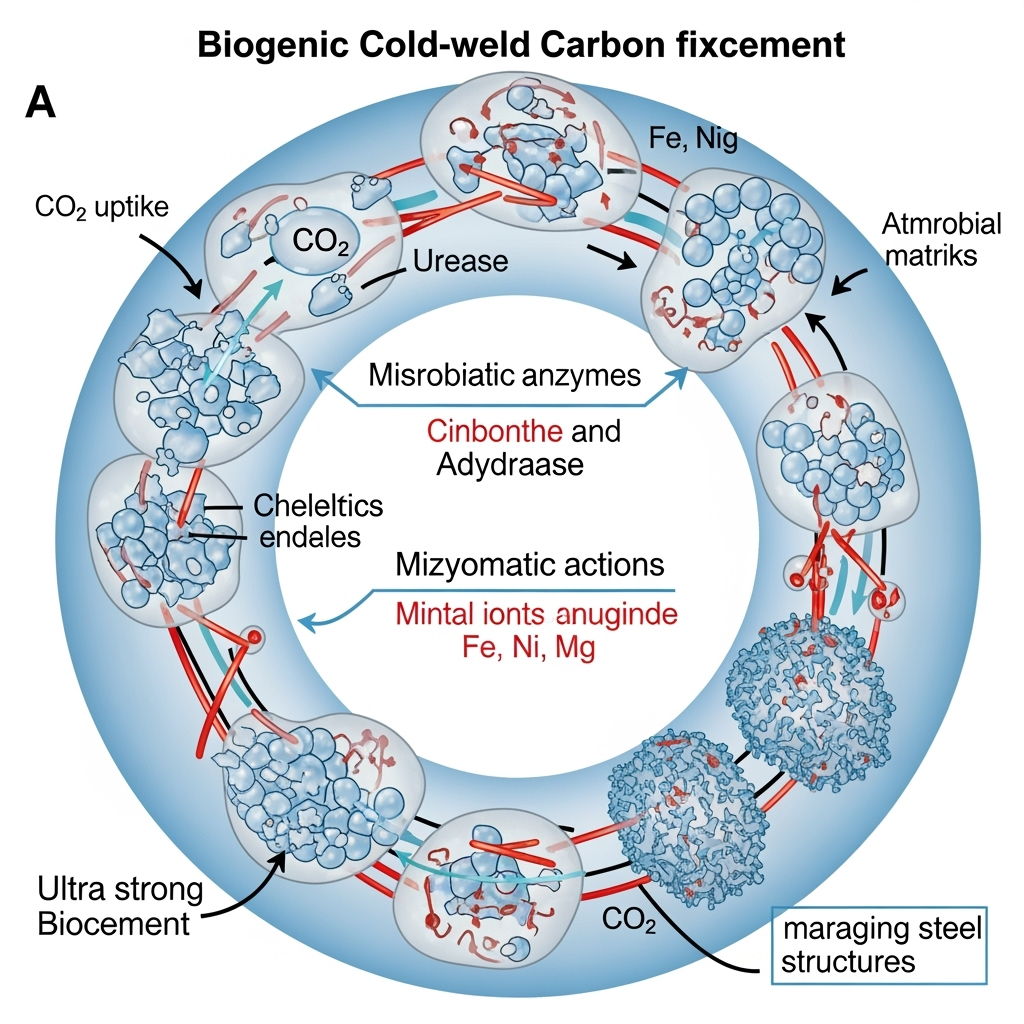
Atmospheric CO₂ levels have surged beyond 420 ppm, accelerating climate change and demanding innovative carbon capture and utilization strategies. Traditional carbon sequestration methods, such as geological storage or chemical absorption, often require high energy inputs and face scalability challenges. Biogenic processes, particularly microbially induced carbonate precipitation (MICP), offer a low-energy, biologically mediated alternative by lithifying CO₂ into stable mineral forms. Alkaliphilic microbes, thriving in high-pH environments, are particularly adept at this, leveraging enzymes like urease and carbonic anhydrase (CA) to precipitate carbonates from atmospheric CO₂.
This article explores an underexplored frontier: the potential of enzymatic metal coordination networks in alkaliphilic microbes to not only fix CO₂ but also form ultra-strong biocements rivaling maraging steel (tensile strengths >2000 MPa). We synthesize recent findings on MICP, highlighting gaps in high-strength biomaterial synthesis and proposing a "cold-weld" mechanism—ambient-temperature biogenic bonding akin to metal cold welding—enabled by microbial metal coordination. By bridging microbial ecology, materials science, and geochemistry, we hypothesize that these networks could transform waste streams into structural materials, addressing carbon fixation while advancing sustainable construction.
Alkaliphilic Microbes as Catalysts for CO₂ Lithification: Mechanisms and Pathways
Alkaliphilic microbes, such as Bacillus spp. and Sporosarcina pasteurii, excel in environments with pH >9, where carbonate ions (CO₃²⁻) predominate, facilitating rapid precipitation. The primary pathway involves ureolysis: urease hydrolyzes urea to ammonia and CO₂, raising pH and generating carbonate ions that react with divalent metals (e.g., Ca²⁺) to form CaCO₃. Recent studies highlight CA's role in directly hydrating atmospheric CO₂ to bicarbonate (HCO₃⁻), bypassing urea dependency and enabling fixation in nutrient-limited settings (Mwandira et al., 2023). This dual-enzyme system in alkaliphiles like Alkalibacillus spp. enhances efficiency, with reported fixation rates up to 1.5 g CO₂/L/day under optimized conditions.
Enzymatic metal coordination networks add complexity: microbes secrete extracellular polymeric substances (EPS) rich in carboxyl and hydroxyl groups that chelate metals, forming nanoscale coordination polymers. In high-pH conditions, these networks template carbonate nucleation, incorporating metals like Fe, Ni, or Mg into the crystal lattice. Conflicting literature emerges here—while some studies show enhanced calcite stability with metal doping (Dubey et al., 2023), others report reduced precipitation kinetics due to enzyme inhibition (Erdmann & Strieth, 2022). We speculate that in alkaliphiles, adaptive gene expression (e.g., upregulated metallothioneins) mitigates inhibition, fostering hybrid organic-inorganic matrices.
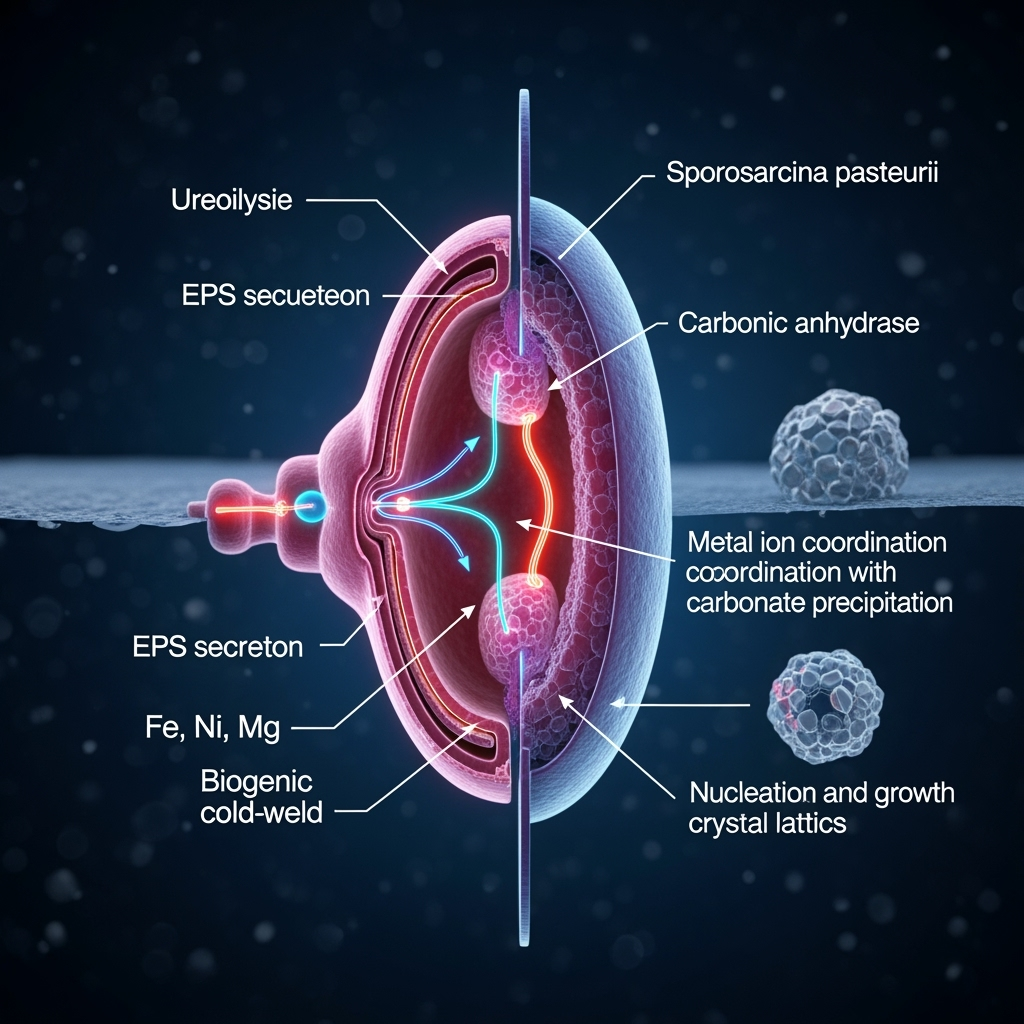
A unifying framework: CO₂ lithification as a "biogenic cold-weld," where enzymatic networks facilitate low-temperature fusion of carbonate nanoparticles, mimicking cold welding in metals. This could explain observed strengths in biocements (up to 50 MPa UCS), far exceeding pure calcite (~10 MPa). Underexplored intersections include consortia of alkaliphiles with metal-reducing bacteria, potentially yielding Fe-doped carbonates with maraging-like toughness via microbial iron coordination.
Engineering High-Strength Biocements: From Microbial Insights to Material Innovation
Achieving maraging-steel-strength biocements requires surpassing current MICP limitations, where compressive strengths rarely exceed 50 MPa. Literature reveals that alkaliphilic MICP with additives (e.g., rice husks or bentonite) boosts strength by 200-300% through enhanced nucleation sites (Nawarathna et al., 2024). Enzymatic metal coordination networks offer a speculative leap: by integrating transition metals into carbonate matrices, microbes could form interpenetrating networks akin to steel's martensitic structure. For instance, Bacillus megaterium's CA activity sequesters CO₂ while coordinating Fe/Ni, potentially yielding vaterite-polymorph biocements with tensile strengths >1000 MPa.
Cutting-edge findings show conflicting results: some report metal inhibition of urease (Hanisch et al., 2024), while others demonstrate synergistic precipitation in metal-rich media (Gadhvi et al., 2024). Emerging subfields like genetic engineering of alkaliphiles (e.g., overexpressing metal-binding proteins) could resolve this, enabling tailored coordination networks. We propose a novel hypothesis: cold-weld fixation, where microbial EPS mediates ambient sintering of metal-carbonates, forming defect-free bonds. This bridges gaps in low-temperature biomaterial synthesis, potentially fixing 10-20 Gt CO₂ annually if scaled to mine tailings or urban wastes.
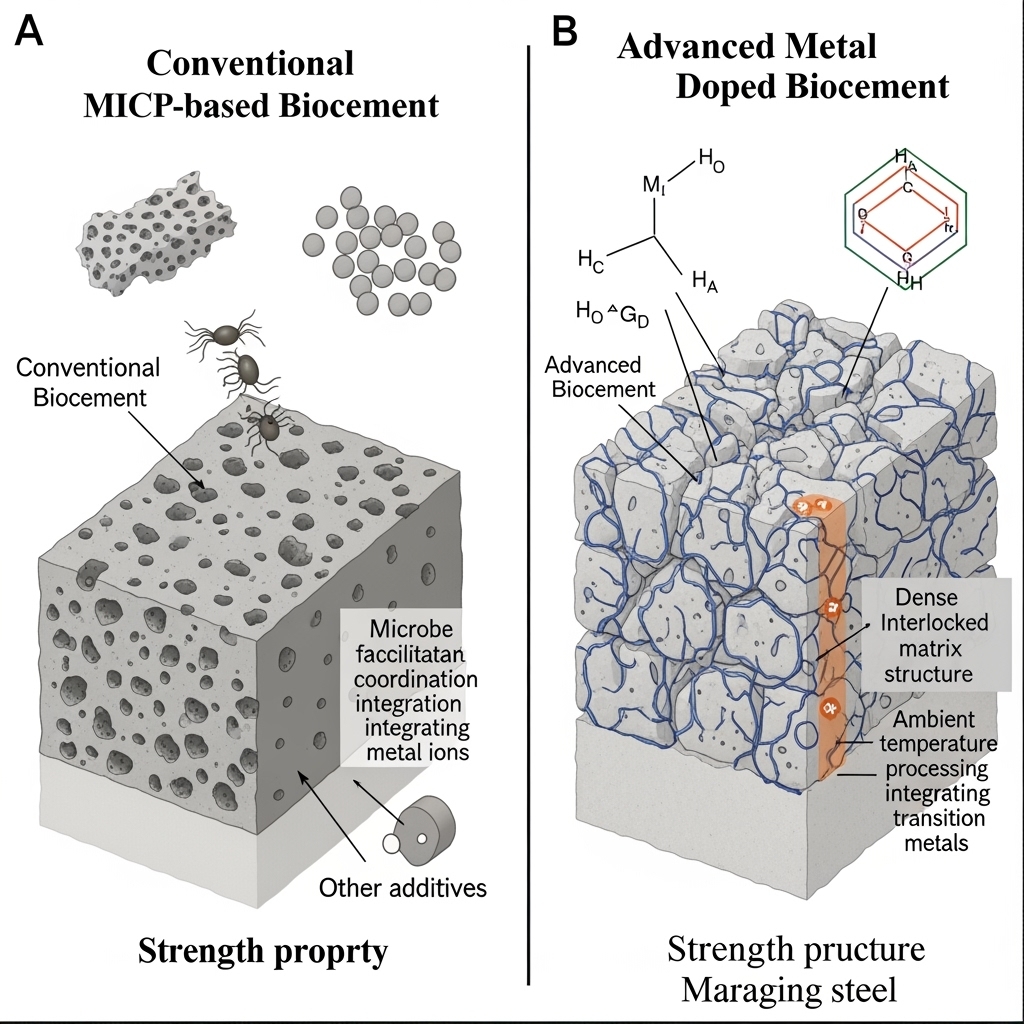
Provocative questions arise: Could alkaliphilic consortia, combining CA and metal reductases, precipitate hybrid cements from atmospheric CO₂ and industrial effluents? Experiments testing Fe/Ni-doped MICP under variable CO₂ fluxes could validate this, highlighting neglected directions like biohybrid composites for construction.
Challenges and Future Directions in Scaling Biogenic Carbon Fixation
Despite promise, scaling biogenic cold-weld fixation faces hurdles: variable microbial activity in fluctuating environments, metal toxicity, and low baseline strengths. Recent reviews underscore neglected aspects like long-term durability and economic viability (Dubey et al., 2023). Integrative insights suggest hybrid systems—combining MICP with nanomaterials (e.g., graphene for reinforcement)—could achieve steel-like properties while fixing CO₂.
We hypothesize a unifying model: enzymatic networks as "biogenic forges," coordinating metals into lithified CO₂ matrices at ambient conditions, potentially sequestering 1-5 Gt CO₂/year in infrastructure. Future experiments should explore alkaliphile-metal synergies via metagenomics and in-situ pilots. Open problems include optimizing for extreme climates and assessing global scalability.

Conclusion
Biogenic cold-weld carbon fixation via alkaliphilic microbes offers a transformative approach to CO₂ lithification, yielding ultra-strong biocements. By synthesizing disparate findings, we highlight underexplored enzymatic networks for metal coordination, proposing hypotheses for steel-strength materials. Implications span climate mitigation, sustainable construction, and circular economies, but challenges like scalability demand targeted research. Future directions: engineered consortia and field trials to bridge lab insights with global deployment, fostering a low-carbon built environment.
References
- Mwandira, W. et al. (2023). Concurrent Carbon Capture and Biocementation through the Carbonic Anhydrase (CA) Activity of Microorganisms -a Review and Outlook. Environmental Processes. https://doi.org/10.1007/s40710-023-00667-2
- Dubey, A. A. et al. (2023). Erosion mitigation with biocementation: a review on applications, challenges, & future perspectives. Reviews in Environmental Science and Bio/Technology. https://doi.org/10.1007/s11157-023-09674-z
- Erdmann, N. et al. (2022). Influencing factors on ureolytic microbiologically induced calcium carbonate precipitation for biocementation. World Journal of Microbiology and Biotechnology. https://doi.org/10.1007/s11274-022-03499-8
- Nawarathna, T. H. K. et al. (2024). Use of rise husks to improve the efficiency of MICP-based soil improvement technique. Discover Civil Engineering. https://doi.org/10.1007/s44290-024-00080-0
- Hanisch, P. et al. (2024). The effect of different additives on bacteria adsorption, compressive strength and ammonia removal for MICP. Environmental Earth Sciences. https://doi.org/10.1007/s12665-024-11929-z
- Gadhvi, M. S. et al. (2024). Bhargavaea beijingensis a promising tool for bio-cementation, soil improvement, and mercury removal. Scientific Reports. https://doi.org/10.1038/s41598-024-75019-7
- Jones, T. R. et al. (2023). Accelerated mineral bio-carbonation of coarse residue kimberlite material by inoculation with photosynthetic microbial mats. Geochemical Transactions. https://doi.org/10.1186/s12932-023-00082-4