Biodegradable Micro-Robots for Targeted Drug Delivery in Chronic Diseases
Chronic diseases such as osteoarthritis, inflammatory bowel disease, and many cancers represent a staggering burden on global health. A primary challenge in their management is the systemic toxicity and off-target effects of potent drugs, which must be administered to the entire body to treat a localized ailment. Micro-robotics offers a paradigm-shifting solution: navigating drug payloads directly to the site of disease. However, the field has been dominated by metallic, externally-powered robots that raise concerns about biocompatibility and require continuous external energy fields (e.g., magnetic) for guidance. A significant gap remains for systems that are fully biodegradable and can operate autonomously within the body by sensing and responding to the specific biochemical cues of their target environment.
This article proposes a new conceptual framework for biodegradable micro-robots that achieve autonomous targeting by integrating three key elements: a chassis built from disease-responsive biomaterials, a bio-hybrid propulsion system guided by inflammatory signals, and an intelligent payload released by logic-gated mechanisms. We synthesize recent advances in hydrogel technology, nanoparticle drug delivery, and cell-based therapeutics to outline a new class of "sense-and-act" micro-robots. These bio-integrated machines could navigate to inflamed tissues, identify pathological hallmarks, and deploy therapeutics in a closed-loop fashion, heralding a new era of precision medicine for chronic diseases.
The Chassis - A Disease-Responsive, Biodegradable Vehicle
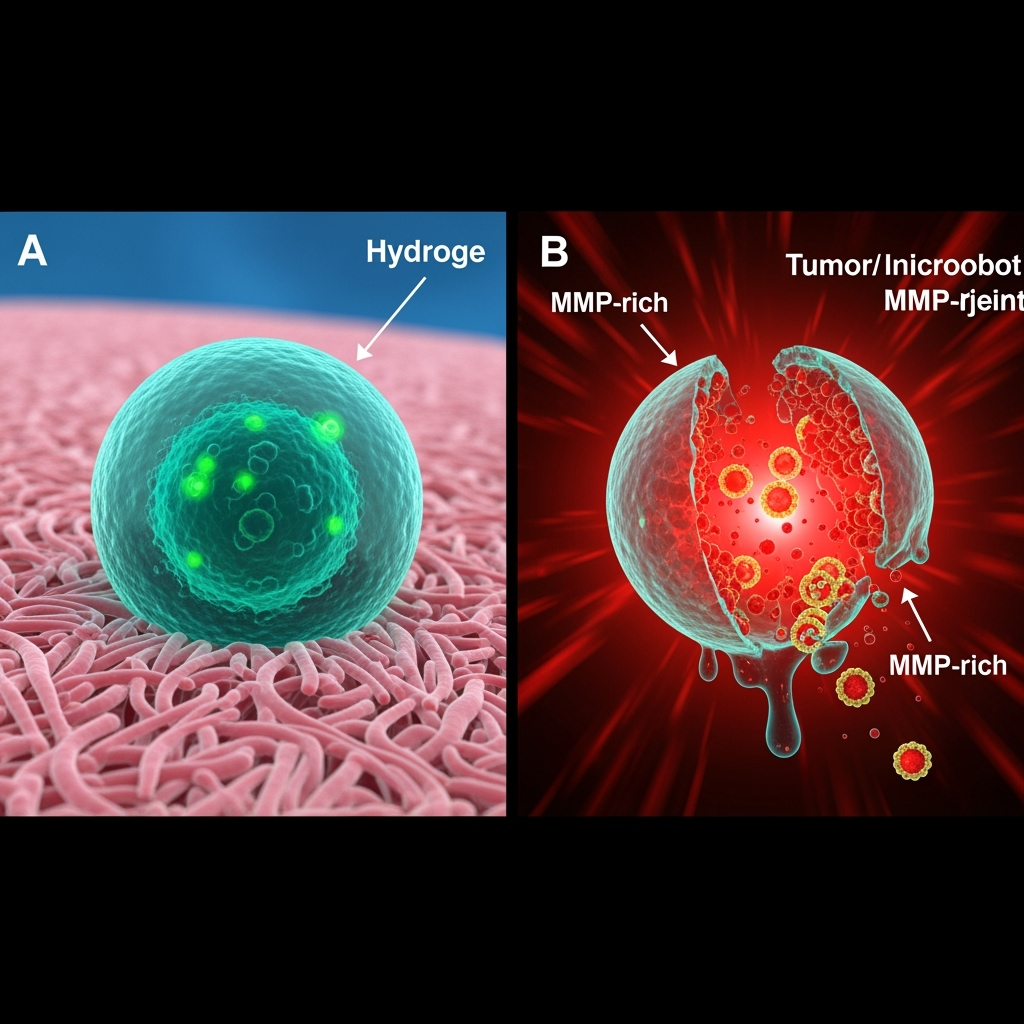
The foundation of an autonomous micro-robot must be a vehicle that is not only biocompatible but also interactive with its environment. Recent advancements in polysaccharide and protein-based biomaterials provide the ideal toolkit. Materials like hyaluronic acid (HA), chitosan, and collagen are not only biodegradable and have low immunogenicity, but their degradation can be programmed to respond to the unique enzymatic fingerprint of a chronic disease. For instance, the synovium in an osteoarthritic joint or the microenvironment of a solid tumor is rich in matrix metalloproteinases (MMPs). A micro-robot chassis built from an MMP-sensitive hydrogel, similar to those being developed for wound healing and regenerative medicine, would be designed for structural dissolution precisely at the target site (Meng, et al., 2025; Zhu, et al., 2025).
This enzymatic degradation is not merely a disposal mechanism; it is an integral part of the targeting and release system. The vehicle's structural integrity would be contingent on its environment. In healthy tissue with low MMP activity, the micro-robot remains intact, sequestering its therapeutic payload. Upon entering a diseased region, the elevated enzymatic activity initiates the chassis breakdown. This ensures that the robot's function is spatially concentrated, minimizing payload release in healthy tissues. Furthermore, as shown in advanced hydrogel designs for diabetic ulcers, these biomaterials can be tuned for specific mechanical properties and porosity, enabling them to carry and protect complex payloads like therapeutic proteins or nanoparticle systems (Se, et al., 2025).
The Engine - Bio-Hybrid Propulsion Towards Inflammatory Signals
A truly autonomous micro-robot must navigate without constant external control. While magnetic and acoustic fields can guide robots, they are cumbersome and non-specific. We propose a bio-hybrid approach that leverages the natural chemotactic capabilities of living cells as an "onboard engine." Chronic diseases create distinct chemical gradients; inflamed tissues release a cocktail of cytokines and chemokines to recruit immune cells. An engineered, non-pathogenic bacterium or even a patient's own neutrophils could be integrated into the micro-robot's chassis to serve as a biological guidance system.
These "engines" would be programmed to seek out specific inflammatory markers. For example, by engineering bacteria to express receptors for tumor necrosis factor-alpha (TNF-α) or specific interleukins, the micro-robot would actively migrate towards the highest concentration of these signals—the heart of the diseased tissue. This transforms the pathological signal itself into a navigational beacon. This strategy mimics the body's own immune response and redirects it for therapeutic ends. While the current literature focuses on the cargo (exosomes, growth factors) rather than the vehicle, the principle of using biological cues for localization is well-established in fields like cancer neuroscience, where neural-tumor crosstalk relies on similar signaling pathways (Sun, et al., 2025).
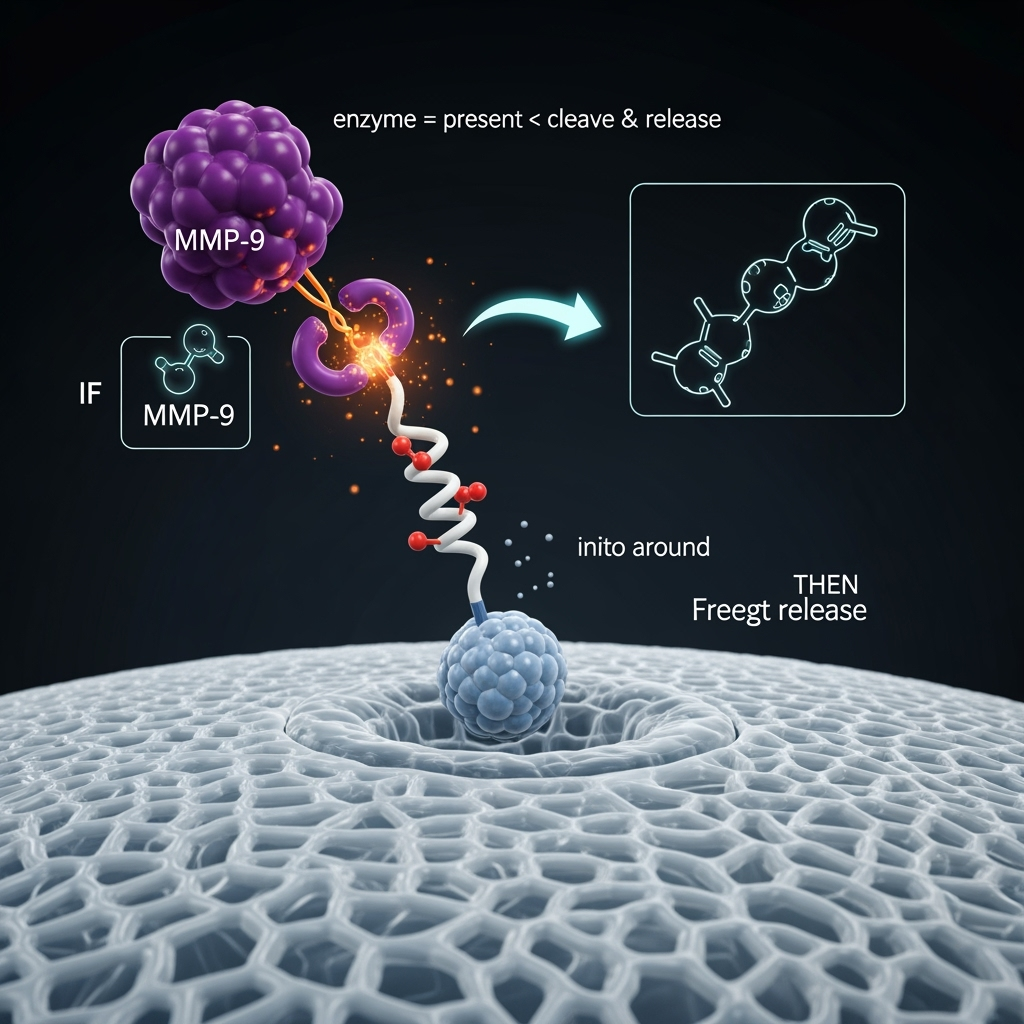
The Payload - Intelligent, Logic-Gated Drug Release
Delivering a drug to the right location is only half the battle; it must be released at the right time and in the correct dose. The proposed micro-robot architecture enables "intelligent" payload release through enzymatic logic gates. Instead of simple diffusion from a hydrogel, therapeutic molecules would be attached to the robot's chassis using linker peptides that are specifically designed to be cleaved by disease-associated enzymes. For example, a potent anti-inflammatory drug could be rendered inactive by tethering it to the hydrogel with a short peptide sequence that is a substrate for MMP-9.
This creates a Boolean logic gate: IF the robot is in a region with high MMP-9 activity (a condition met at the disease site), THEN the linker is cleaved and the drug is released. This ensures that the therapeutic action is a direct consequence of the pathological state. This approach builds on work showing the efficacy of nanoparticle systems, such as niosome-dendrimer platforms, in enhancing drug cytotoxicity at the target site (Kaveh Zenjanab, et al., 2025). By combining the targeting of a chemotactic engine with the logic-gated release from a biodegradable chassis, the system achieves a high degree of precision. Furthermore, multiple drugs could be attached with different enzyme-sensitive linkers, allowing for combinatorial therapy that is responsive to the complex enzymatic landscape of the disease.
Conclusion
The convergence of biodegradable materials, bio-hybrid systems, and enzyme-responsive drug delivery creates a pathway toward fully autonomous, intelligent micro-robots for treating chronic diseases. This article puts forward a conceptual framework for a system that navigates using the chemical signals of inflammation, identifies its target via specific enzymatic activity, and performs its therapeutic action by releasing a payload in a logic-gated manner before degrading into harmless byproducts. This represents a paradigm shift from externally-powered, permanent hardware to self-guided, transient biological machines.
Significant challenges remain before this vision becomes a clinical reality. The potential immunogenicity of bio-hybrid systems must be carefully managed, the long-term stability and navigational accuracy of the cellular "engines" need to be established, and the manufacturing of these complex, multi-component systems must be made scalable. However, the foundational technologies are already emerging. The development of advanced hydrogels, precision nanoparticle drug carriers, and our growing understanding of the molecular signals of disease provide the necessary building blocks. By integrating these fields, we can begin to design and test the first generation of biodegradable micro-robots that act less like machines and more like intelligent, artificial cells programmed to hunt and heal.
References
- Abaza, T., Mohamed, E. E., & Zaky, M. Y. (2025). Lipid nanoparticles: a promising tool for nucleic acid delivery in cancer immunotherapy. Medical Oncology. https://doi.org/10.1007/s12032-025-02939-3
- Cavallo, J., & Kasapis, S. (2025). Quantifying Microstructure to Better Control Bioactive Compound Delivery in Protein-Based Systems from Low- to High-Solid Preparations for Improved Human Health. Food Biophysics. https://doi.org/10.1007/s11483-025-10011-w
- Fang, F., et al. (2025). Collagen-based biomaterials in neural injury repair: current advances and future perspectives. Collagen and Leather. https://doi.org/10.1186/s42825-025-00209-6
- Kaveh Zenjanab, M., et al. (2025). Dual-loaded niosome-dendrimer nanoplatform enhances Tirapazamine delivery to hypoxic breast cancer cells. Scientific Reports. https://doi.org/10.1038/s41598-025-14704-7
- Meng, Y., et al. (2025). Exo-hydrogel therapy: a revolutionary approach to managing diabetic complications. Journal of Nanobiotechnology. http://dx.doi.org/10.1186/s12951-025-03621-6
- Mittal, D., & Verma, A. K. (2025). Nanotherapeutic interventions to enhance cisplatin sensitivity in ovarian cancer cells. Egyptian Journal of Medical Human Genetics. https://doi.org/10.1186/s43042-025-00756-x
- Radha, R., et al. (2025). Cellulose-based materials for antimicrobial water treatment: functionalization strategies and applications. Discover Applied Sciences. https://doi.org/10.1007/s42452-025-07558-1
- Se, A. M., Li, L., & Yu, M. (2025). Poloxamer-based hydrogel with EGCG and rhEGF for diabetic foot ulcer treatment. Journal of Materials Science: Materials in Medicine. http://dx.doi.org/10.1007/s10856-025-06917-z
- Sun, H., et al. (2025). Integrating neuroscience and oncology: neuroimmune crosstalk in the initiation and progression of digestive system tumors. Molecular Cancer. http://dx.doi.org/10.1186/s12943-025-02412-9
- Zhu, Z., et al. (2025). Carrier rocket-inspired hydrogel microspheres targeting subchondral bone osteoclast activity alleviate osteoarthritic pain and cartilage degeneration. Journal of Nanobiotechnology. http://dx.doi.org/10.1186/s12951-025-03598-2